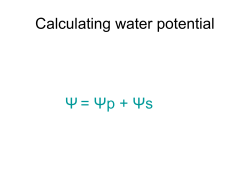Solutions
Name:_________________________ Per:_______ Solutions 1. A homogeneous mixture of particles in a dissolving medium is called a _______________. 2. The substance dissolved is called the ____________ and the medium in which it is dissolved is called the _____________. 3. Circle the solute and underline the solvent. a) b) c) d) Salt water Sugar water A solution of 20% HCl and 80% water Rubbing alcohol: 60% alcohol, 40% water 4. The series of diagrams above show NaCl dissolving in water. Explain what is happening. __________________________________________________________________________________________ ________________________________________________________________________________ 5. TRUE or FALSE (if false, correct it so that it’s true) After salt completely dissolves in water, it will eventually settle to the bottom of the container if it is left undisturbed. 6. The series of diagrams above show sugar (C12H22O11) dissolving in water. Explain what is happening. __________________________________________________________________________________________ __________________________________________________________________________________________ Name:_________________________ Per:_______ 7. List three things you could do to increase the rate of dissolving of a solid solute in a liquid solvent. ____________________ ____________________ ____________________ 8. List three things you could do to increase the rate of dissolving of gas in a liquid solvent: ____________________ ____________________ ____________________ 9. Define “solubility”: _______________________________________________________________________________ _______________________________________________________________________________. 10. Match each term with the correct description: ___ saturated A. solution with less than the max amount of solute dissolved ___ supersaturated B. solution with more than the max amount of solute dissolved ___ unsaturated C. solution with exactly the max amount of solute dissolved 11. You have a clear solution of sugar water. You add a sugar crystal to the solution and….. a) it sinks to the bottom and does not dissolve. The solution is saturated / unsaturated / supersaturated b) it causes many crystals of sugar to form in the solution. The solution is ______________________. 12. Three factors that affect solubility are: ____________________ ____________________ ____________________ 13. High temp. makes ______________ less soluble, but make _____________ more soluble. 14. Why do sodas left open out of the fridge go “flat”? __________________________________________________________________________________. 15. a) In general, what types of substances dissolve well in water? Ionic / Polar / Nonpolar b) Why?______________________________________________________________________________ 16. What phrase is used to describe which solutes dissolve in which solvents? ______________ dissolves _____________ 17. Which mixtures contain a solute that would dissolve in the solvent? (circle all that apply) A. polar sugar molecules (C12H22O11) in polar ethanol (C2H5OH) B. polar dichlorodifluoromethane (or Freon) (CCl2F2) in nonpolar carbon tetrachloride (CCl4) C. polar ethanoic acid (CH3COOH) in water (H2O) 18. Circle A. B. C. the letter of each sentence that is TRUE about aqueous solutions. When a solution is filtered, both solute and solvent will pass through the filter paper. Ionic compounds and polar covalent compounds dissolve easily in water. Nonpolar covalent compounds (like oil, grease, and gasoline) dissolve easily in water.
© Copyright 2026