Corticosteroid modulation of interleukin-1 hematopoietic effects and
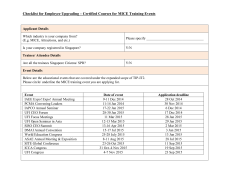
From www.bloodjournal.org by guest on November 20, 2014. For personal use only. 1994 84: 1457-1463 Corticosteroid modulation of interleukin-1 hematopoietic effects and toxicity in a murine system J Rinehart, EW Delamater, L Keville and J Measel Updated information and services can be found at: http://www.bloodjournal.org/content/84/5/1457.full.html Articles on similar topics can be found in the following Blood collections Information about reproducing this article in parts or in its entirety may be found online at: http://www.bloodjournal.org/site/misc/rights.xhtml#repub_requests Information about ordering reprints may be found online at: http://www.bloodjournal.org/site/misc/rights.xhtml#reprints Information about subscriptions and ASH membership may be found online at: http://www.bloodjournal.org/site/subscriptions/index.xhtml Blood (print ISSN 0006-4971, online ISSN 1528-0020), is published weekly by the American Society of Hematology, 2021 L St, NW, Suite 900, Washington DC 20036. Copyright 2011 by The American Society of Hematology; all rights reserved. From www.bloodjournal.org by guest on November 20, 2014. For personal use only. Corticosteroid Modulation of Interleukin-l Hematopoietic Effects and Toxicity in a Murine System By John Rinehart, Elizabeth W. Delamater, Lisa Keville, and John Measel Interleukin-l (IL-l)has been shown to ameliorate the hematopoietic toxicities of antitumor chemotherapeutic agents in both mice and humans. However, IL-l toxicity in humans is considerable and is similar to the systemic inflammatory toxicities induced by IL-3,IL-6, and other cytokines with pleiotropic biologic activities, eg, fever, nausea, malaise,and hypotension. We hypothesized that corticosteroids may reduce IL-l toxicity without reducing IL-l hematopoietic effects in vivo. C3H/HeJ mice (female, 6 weeks) were treated for 7 days subcutaneously with cortisone acetate (CA), (0.1. 0.25, or 0.5 mg/d/mouse), intraperitoneally with IL-l (1or 2 pg/d/mouse), or both. Asexpected, IL-l increased white blood cell counts, splenic granulocyte-macrophage colonyforming units, and spleen cell number, and protected mice fromlethal dosesof carboplatin (200 mg/kg; Paraplatin, Bristol Laboratories, Evansville, IN) administered the day after completion of the 7 days of IL-l administration. CA did not significantly block the hematopoietic effects of IL-l or the ability of IL-l to protect mice from the hematopoietic toxicity of carboplatin. IL-l administered to mice at 8 pg/d/ mouse for 5 days induced decreased activity, roughening of hair, diarrhea, pancytopenia, multiple metabolic abnormalities, and death in 60% of mice. IL-l at thetherapeutic doses (0.5 to 2 pg/d) was not toxic. CA in a dose-dependent manner blocked all of the above mentioned toxicities when administered 24 hours and 30 minutes before each IL-l injection. CA also decreased IL-l-induced increase in plasma tumor necrosis factor levels at the time pointexamined. 0 1994 by The American Societyof Hematology. H postorbital venous plexus using a microcapillary tube coated with 5% EDTA in Hanks' balanced salt solution (HBSS; GIBCO-BRL, Gaithersburg, MD). Sixty microliters of the blood was diluted into 120 pL ofEDTA solution andthewhiteblood cell (WBC) and platelet counts were determined using a model S+IV B Coulter counter (Coulter Electronics, Hialeah, FL). Smears were made of the blood samples and stained with Wright's stain (Sigma Chemical CO,St Louis, MO). These slides were examined by light microscopy and the percentages of lymphocytes. granulocytes, and monocytes were determined. Preparation of spleen mononuclear cell suspensions and leukocyte-conditioned media (LCM). Animals were killed by cervical dislocation, their spleens removed and placed in cold Iscove's modified Dulbecco's Eagle's Medium (IDMEM; GIBCO). Spleens were disassociated by pressing through a Cellector tissue sieve (PGC Scientifics, Gaithersburg, MD) into 60-mm tissue culture dishes (Falcon 1007, Becton Dickinson Labware, Lincoln Park, N3) containing IDMEM supplemented with 20% fetal calf serum (FCS). Cells were layered on Ficoll-Paque (Pharmacia Diagnostics, Silver Spring, MD) in 15-mL polypropylene tubes. These were centrifuged at 400g for 25 minutes and interface cells were harvested. Spleen mononuclear cells were washed three times by centrifugation at 200g. Spleen mononuclear cells from treated animals were cultured in a methylcellulose hematopoietic progenitor cell assays for CFU-GM (see below). To produce LCM, washed spleen mononuclear cells from normal, spleens from untreated animals were resuspended in IDMEM + 20% FCS + 10 yg/mL phytohemagglutinin to a final concentration EMATOPOIETIC growth factors and cytokines have shown effectiveness in reducing the toxicity of anticancer chemotherapeutic agents. Both granulocyte colonystimulating factor and granulocyte-macrophage colony-stimulating factor (GM-CSF) have been shown to reduce the period of granulocytopenia if given after administration of chemotherapeutic agents that induce damage to hematopoietic precur~ors."~ Other cytokines with activity on early hematopoietic progenitors have been tested in animals or clinical trials and have the potential to reduce suppression by chemotherapeutic agents of platelet production, and production and function of various lymphocyte subsets, eg, interleukin-l (IL-I), IL-3, IL-6, IL-10, IL-l l , stem cell growth factor, and other^.^"' Many of the cytokines with pleiotropic biologic activities (eg, IL-l, IL-3, and IL-6) that act on early hematopoietic precursors share similar systemic toxicities: fever, chills, malaise, nausea, rashes, hypotension, and fluid retention. Preliminary data suggest that some of these toxicities may be caused by a secondary release of tumor necrosis factor (TNF), tumor growth factor (TGFP), and other cytokines which may also negate the positive hematopoietic effects of the administered ~ y t o k i n e . ' ~ " ~ We have shown in vitro that corticosteroids enhance GMCSF, IL- 1, and IL-3 induced formation of granulocyte-macrophage colony forming units (CFU-GM) and inhibit IL-1induced release of TNF by human mononuclear cells.16 We hypothesized that corticosteroids may block IL- 1 toxicity and enhance IL-I hematopoietic effects in vivo. We report the experiments examining this hypothesis in a murine model. MATERIALSAND METHODS Mice. Female C3WHeJ mice were obtained from theAnimal Production Area, National Cancer Institute-Frederick Cancer Research and Development Center (Frederick, MD). Animals were routinely maintained at 5 to 8/cage inside filter bonnets in an isolation room, with food and water provided ad libitum. Mice were quarantined for 1 week before treatment. Experiments were performed using mice at 5 to 8 weeks old. Preparation of peripheral blood smears for differential counts. Animals were anesthetized by Forane (isoflurane) inhalant (Anaquest, Madison, WI), thenblood samples were obtained from the Blood, Vol 84, No 5 (September l), 1994:pp 1457-1463 From the Divisions of Hematology and Oncology, the Department of Medicine, Scott & White Clinic & Memorial Hospital: the Scott, Sherwood and Brindley Foundation; andTexasA&M University Health Science Center, College of Medicine, Temple, TX. Submitted September 7, 1993: accepted April 29, 1994. Supported by National Institutes of Health Grant No. R01 CA51160-011A and institutional funds from the Scott, Sherwood and Brindley Foundation and Texas A & M University Health Sciences Center. Address reprint requests toJohn Rinehart, MD, Scott & White Clinic, 2401 S 31st St, Temple, TX 76508. The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked "advertisement" in accordance with 18 U.S.C. section 1734 solely to indicate this fact. 0 1994 by The American Society of Hematology. 0006-497//94/8405-00$3.00/0 1457 From www.bloodjournal.org by guest on November 20, 2014. For personal use only. 1458 of 6 X 1Oh/mL. Thecellsuspension was placed in 25-cm2 tissue culture flasks, then incubated at 37°C for 5 days. After incubation, thecellsupernatant(ie,LCM)wascentrifugedandtheresultant supernatant was passed through a 0.22-pm syringe filter (Gelman Sciences, Ann Arbor, MI), then stored in 2-mL aliquots at -4°C. Preparation and cultureof bone marrow (BM). Mice were killed by cervical dislocation, and tibias placed in chilled HBSS. BM cells were obtained by flushing cold HBSS through tibiaswith a 27-gauge needle. Cells were washed once in cold HBSS, then resuspended to 3 X lo6 cells/mL in IDMEMsupplemented with 20%FCS. BM a mononuclear cells (3x10') were incubated in methylcellulose at final concentrationof 1% supplemented with 20%FCSand l% antibiotic-antimycoticsolution.Positivecontrolcultures had 10% LCM added. The cell cultures were plated in triplicate wells in 6well tissue culture dishes and incubated at37°C in a fully humidified atmosphere of 5% COz for 7 days. On the seventh day, the number of CFU-GMweredetermined by examinationusing an inverted microscope. Administration of' biologic response modifiers or rhemotherupy. IL-I,f? was a gift from Syntex Discovery Research (Palo Alto, CA). IL-IP was dissolved in sterile saline containing 0.05% bovine serum albumin (BSA); injections (0.2 mL/mouse) were administered intraperitoneally (IP). Cortisone acetate (CA)(Cortone; Merck, Sharp and Dohme, West Point, PA) was dissolvedin sterile IDMEM; injections (0.2 mL/mouse) were administered subcutaneously (SQ). Carboplatin (Paraplatin, Bristol Laboratories, Evansville, IN) was disto package direcsolved in sterile 0.9% NaCl for injection according tions. Individual animals in an experimental group were administered an intravenous (IV) injection of Carboplatin (0. I mL) in the lateral or dorsal tail vein. Cell proliferation. Cells were prepared as described in previous sections, then resuspended to I X IO6 cellslmL in RPMI-1640 containing 20% fetal bovineserum, supplemented with 100 pg/mL penicillin, 0.1 pg/mL streptomycin, 0.25 pg/mL amphotericin-B, and 0.2 mmol/L L-glutamine. The cell suspension was pipetted into quadruplicate wells of a 96-well round-bottomed tissue cultureplate in 100 pL. Proliferation was induced by human recombinant IL-2. A twofold serial dilution of IL-2(10,000 to 156 Cetus U/mL) was prepared and100 pL wasaddedtotheappropriate wells. The plates were incubated at 37"C, 5% COz for 72 hours. After incubation, the wells were pulsed with 0.5 pCi 'H-thymidine, for 4 hours. The cells were harvested using a PHD multiple automated sample harvester (Cambridge Technology Inc, Cambridge, MA) and the radioactivity on theglass filter discsobtainedfromtheharvest was measured by as meancountsper liquidscintillation.Thedatawereexpressed minute (cprn) from quadruplicate wells of 5,000 the U/mL concentration that induced the highest cpm in all experiments. Staining of BMor spleen forflow, cytometty. BM cells and spleen mononuclearcellswereobtainedasdescribed above. Cellswere X IO' cells/mL washed three times in HBSS and resuspended to 2 in HBSS without phenolred containing I % BSA (Fraction V, Sigma) (staining buffer). Ethidium bromide (5 mg/mL in I O mmoUL TRIS, 1 mmol/L EDTA) was added to the cell suspension. After incubating 20 minutes in an ice bath, the cells were washed by adding 2 mL of staining buffer and pelleting at 300g for 6 minutes at 4°C. Stained cells were resuspended in 400 pL cold staining buffer and held on ice until quantitation by a flow cytometer. Enzyme-linked immunosorbent assay (ELISA). ELISA was used to determine the concentration of putative inhibitors of hematopoiesis. Human TNFa was assayed using a commercially available kit (Predicta;GenzymeCorp,Cambridge,MA).Theabsorbancewas measured at 450 nm. Concentration of TNFa was determined by comparing results of test samples with a standard curve generated by the same assay. The sensitivity of this assay is 10 pg/mL. Statistical methods. Statistical analysis was performed using the RINEHART ET AL 12. t t IL-l 1.0pg IL-l 1.0 pg + CA 0.1 mg "E 10- E " .8X 8a 4 61 / l 2 04 0 2 4 6 128 10 Days t t CA 0.1 rng IL-l 1.0 pg 2i 04 0 l 2 4 6 8 10 12 8 10 12 Days 16, t 0 Control IL-l 1.0 pg 2 4 6 Days Fig1. Six-week-old female C3HIHeJ mice were injected with IL1 IIP at 1.0 pglmouse) or CA IS0 at 0.1 mglmouse) or both IL-l and CA. Controls were injected with carrier IP and SO for 7 days 16 mice/ group). Mice were bled from the retro-orbital plexus and peripheral blood counts w w e determined as described in Materials and Methods. A, AGC; B, ALC; C, platelets. From www.bloodjournal.org by guest on November 20, 2014. For personal use only. INTERACTIVE EFFECTS OF IL-l AND CORTICOSTEROIDS 1459 Table 1. Effect of CA and IL-lp on Hematopoietic Parameters in C3H/Hd Murine Spleens Dose (mouseld X 7 d)* IL-l 2 pg Control Spleen weight (mg) No. mononuclear cells/spleen ( ~ 1 0 ' ) Spleen CFU-GM* YO Spleen leukocytes in S-phase Spleen leukocyte proliferation in response to IL-2 153 16.3 807 2.5 1,745 t 10t t 3.7 2 119 t 0.4 t 41711 CA 0.25 mg IL-l 2 pg 52 I 35 8.1 t 0.6 61 -t 249 0.7 t 0.15 455 t 46 416 t 155 137 t 12.45 36,696 -t 5,4141 14.9 t 1.65 15,312 t 1,6329 + CA 0.25 mg 391 128.5 74,422 18.3 14,672 t 365 t 0.25 f 16,6639 t 0.85 t 8415 Mice were killed on day 8 after 7 days of SQ CA, IP IL-Ip, or both at doses indicated. ? SD (n = 15 to 17 mice, three experiments). Numbers are total CFU-GM/spleen. 5 P < .01 compared with control. 11 Data presented as mean cpm t SD; IL-2 concentration = 5,000 Cetus U/mL. t Data presented as mean + Student's r-test and one-way analysis o f variance. Differences i n survival betweenvarious treatment groups were analyzedusing Fisher's test. x* RESULTS In initial experiments, we administered IL-1 either at 1.0 or 2.0 pg/d/mouse IP, CA at 0.1 or 0.25 mg/d/mouse SQ or both IL-1 and CA for 7 days. Controls received injections both IP and SQ of appropriate carriers. Peripheral blood counts were determined the day before, during, and after treatment as indicated in Fig1. IL-1 alone suppressed the absolute lymphocyte count (ALC), the absolute granulocyte count (AGC), and platelet count during treatment, but the values of these parameters increased above baseline and control levels by day 12. CA alone at 0.1 mg/d/mouse had no significant effects on these parameters, but CA administered in combination with IL-1 attenuated the decrease in AGC, ALC, and platelet count seen during IL-l administration alone, and attenuated the rebound in these parameters seen when IL-1 was administered alone. The effect of these treatments on splenic parameters is seen in Table 1. Mice were killed the day after completing the treatments as described above and in Table 1. IL- 1 alone increased splenic weight, cell number, CFU-GM, the fraction of cells in S-phase, and proliferative response to IL-2. CA decreased all of these parameters when administered alone. However, when CA was administered with IL-1, splenic CFU-GM increased in number compared with IL- 1 alone. The effect of these treat- ments on BM parameters is presented in Table 2; CA, ILl, and the combination did not significantly change the number of cells or CFU-GM /femur or the fraction of cells in Sphase. In the next series of experiments, we examined the protective effects of IL-1 alone (2 ,ug/d/mouse), CA alone (0.1, 0.25, or 0.5 mg/d/mouse), and IL-l plus CA when given for 7 days before carboplatin, 200 mgkg; 20/24 control mice died compared with 0/20 mice after receiving IL-1 alone ( P < .01) (Fig 2). IL-1 plus CA was as effective as IL-1 alone in rescuing mice from carboplatin toxicity when all three doses of CA administered with IL-1 were analyzed together (1/28 died) and compared with IL-1 alone, or when the three IL-I plus CA groups were separately compared with IL-l alone (see Fig 2). Interestingly, CA alone at 0.5 mg/d/mouse also improved post-carboplatin survival ( P < .01). IL-1 and IL-1 plus CA reduced, but did not eliminate, the marked decrease in AGC, ALC, and platelet counts induced by Carboplatin, 200 mgkg (Fig 3). To determine if CA reduced toxicity of IL- 1, weexamined the toxicity of increasing doses of IL-1: 2 pgldmouse induced no apparent toxicity by inspection of mice; 4 and 6 pg/d/mouse induced weight loss, roughened fur, diarrhea, and decreased activity, but no deaths (data not shown). Mice treated with 8 pg/d/mouse for 5 days died (6/10) or were moribund by day 6 (Table 3). Administration of CA 24 hours and then30 to 60 minutes before each K- 1 injection reduced mortality in a dose-dependent manner; no deaths were ob- Table 2. Effect of CA and IL-lp on Hematopoietic Parameters in C3HIHe.J Murine Bone Marrow Dose Imouseld Control No. bone marrow cells/femur ( ~ 1 0 ' ) Bone CFU-GM marrow % Bone marrow cells in S-phase Bone marrow cell proliferation in response to IL-2 6.9 526 581 12.7 9,391 t 0.9t t 96$ t 0.3 t 1,0615 * Numbers are total CFU-GM/femur. 5 Data presented as mean cpm t SD; IL-2 concentration = 5,000 Cetus U/mL. d)* CA 0.25 mg IL-l 2 pg IL-l 2 pg + CA 0.25 mg 7.3 t 0.6 618 t 150 10.9 14.6 I 0.4 13,627 t 1,114 6.4 t 0.6 532 -t 51 13.3 t 0.8 10,378 t 2,148 5.6 t 0.6 t 146 t 0.8 8,302 t 452 Mice were killed on day 8 after 7 days of SQ CA, P; IL-l@,or both at doses indicated. t Data presented as mean +- SD (n = 15 to 17 mice, three experiments). X From www.bloodjournal.org by guest on November 20, 2014. For personal use only. 1460 A - RINEHART ET AL ment with CA. CA aloneatthehigher doses induced expected modest hematologic and biochemical alterations. but mice receiving CA alone appeared well and no deaths were observed (Table 3, and Figs 4 and S). 100 80 .-I g DISCUSSION 60 The data presented here support the hypothesis we developed; in vivo corticosteroids may blockthetoxic effects, 2 al g 40 Control (24) CAO.lmg (10) t CA 0.25 mg (10) -+- CA 0.5 mg (8) -a- 20 & t Control t 0 2 4 6 8 1'0 14 1'2 1 Days " I 8 CA0.25mg t IL-l 2pg t CA 0.25 mg + IL-l 2 pg 4 - 80 2 0- , , , , , -2 0 2 4 6 8 m , , , , , , 10 12 14 16 18 20 Days c al G 40- -" IL-l 2 ug (20) a 20 - 8 t IL-l 2 ug + CA O.lmg (10) t IL-l 2 ug + CA 0.25 mg (10) A IL-l 2 ug + CA 0.5 mg (8) 6 0 04 0 B 2 8 4 6 1 0 1 2 1 4 Days E E '0 z 4 X Fig 2. In three separate experiments, 6-week-old female C3H/HeJ mice were injected with IL-l (IP at 2 pgldlmouse), CA (SO at 0.10, 0.25, or 0.5 mgldlmouse) or both IL-l and CA or carrier only SO and IP for 7 days (-7 through -1). On day zero, mice were administered 200 mglkg carboplatin IV. Mice were then followed daily for survival without further manipulation. The number of micelgroupare as indicated in parenthesis. (In Fig 28, all of the lines except IL-l 2 Ng+CA 0.5 mg are superimposed.) served in animals receiving CA 0.5 mg/d/mouse plus IL-l. Preadministration of CA also reduced weight loss induced by IL-1 and improved activity levels in surviving mice. IL1 at this toxic dose induced multiple hematologic and biochemical abnormalities (Figs 4 and 5); by day 6, total WBC count,AGC,ALC,and platelet countwere allmarkedly reduced. (Compare the"zero", ie, no cortisone controlmice between Fig 4A (noIL- 1, no CA) and Fig 4B(IL- l , no CA). CA partially reversed IL- l-induced thrombocytopenia and neutropenia and, infact, increased AGC above controllevels. Asexpected,thisschedule of IL-1also increasedplasma serum glutamic oxaloacetic transaminase, andblood urea nitrogen (BUN)and markedlydecreasedplasma glucose, iron, and cholesterol. Mice pretreated with CA showed ameliorization of these IL- l-induced hematologic and biochemical abnormalitiesin a dose-dependent manner. IL- 1 administration at 8 pg/d/mouse(Table 3), but not 2 pg/d/mouse, induced high plasma TNFwhich were decreased by pretreat- sa 2 r : 0-2 - t Control t -t 2 0 4 6 8 10 12 CA 0.25 mg IL-l 2pg CA 0.25 mg + IL-l 2 pg 14 16 18 4 20 22 20 i 22 Days 12- C "c 10~ , "-2 d --f 2 4 6 Control CA 0.25 mg 8 12 10 l4 18 16 Days Fig 3. Mice (8/group) were treatedas described in the figure legend for days -7 through -1. All mice received carboplatin 200 mglkg on day 0. Mice were bled on indicated days t o determine peripheral blood counts. A,AGC; B, ALC; and C, platelets. From www.bloodjournal.org by guest on November 20, 2014. For personal use only. INTERACTIVEEFFECTSOF 1461 IL-l ANDCORTICOSTEROIDS Table 3. Survival and Plasma TNF Levels After High-Dose IL-1/3 in C3H/He.J Mica: Effect of Treatment With Cortisone Acetate No. DeathsFotal Control CA 0.25 mg CA0.5 mg lL-l* IL-l CA0.1 mg IL-l CA 0.25 mg IL-l CA 0.5 mg + + + 016 013 013 420 6110t 216 2110 0110 Plasma TNF Levels (pg/rnL) 0 0 0 ? 153$ 0 0 0 * C3HlHeJ mice received no IL-10 or IL-10 at 8 pglmouseld ondays 2,3,4, and 5 without or with CA, SQ on days 1,2,3,4, and 5. CA was given 30 to 60 minutes before IL-10. t Treated animals died on day 5; on day 6 surviving animals were exsanguinated by cardiac puncture and TNFo levels determined by ELISA. Data presented as mean 2 SD of the mean. Two separate experiments were performed. * but not the hematopoietic effects of IL-1.Thus, this study surprisingly showed that CA blocks some immunologic effects of IL-1while sparing the examined hematologic effects in vivo, and therefore, supports our previously reported in vitro observations.’6 Corticosteroids induce multiple metabolic and immunologic effects when administered in pharmacologic doses to mice and Just as complex are the protean effects induced by administration of IL-1.” Although our present work mayprovide some clues, the mechanisms of these interactions are of undoubted complexity and will require additional studies to elucidate. IL-I at toxic doses induced measurable plasma TNF levels, and pretreatmentwith CA blocked development of IL-1 toxicity and reduced the observed levels of TNF below our assay level of detection. TNF may induce many of the toxicities associated with IL-1administration in humans and mice; eg, TNF can induce hypotension, fluid loss into tissue with resultant hypovolemia and increase in BUN, as well as WBCand platelet margination, nausea, and other systemic symptom~.’’,’~ TNF may also decrease proliferation of hematopoietic progenitor^.'^ However, in our studies, the lowest dose of CA used completely eliminated the increased TNF levels at the single time point examined, but not all IL-1 toxicity. Several possibilities may explain this paradox; local TNF release and action maybe important in TNF toxicity and measurement of plasma levels may underestimate TNF effects. Furthermore, we only obtained TNF levels in plasma at one time point that may not have been optimal in reflecting TNF release. Thus, TNF may have been released but undetected at the time point examined. Finally, other cytokines may play a role and may act alone or synergistically with TNF at levels below our levels of detection to induce toxicity observed. For example, IL-6 and interferons may induce inflammatory-related systemic effects as well as TNF. IL-1 may induce toxicity unrelated to induced release of cytokines; IL-1 may directly enhance margination of oxidantreleasing granulocytes and macrophages into sensitive tissues. The decreased WBC counts observed with toxic doses of IL-1 supports this possibility. Finally, IL-I may directly induce alteration in vascular endothelial cells to ultimately induce tissue hypoperfusion.” CAdidnot block the hematopoietic effects of IL-I in these studies and, in fact, appeared to enhance IL- 1 induction of splenic CFU-GM. We have not explained as yet the mechanism of this latter effect. Moreover, these results must be regarded with caution because we examined only one time point and one dose of CA (Table 1). Nevertheless, we did examine multiple doses of CA in combination with IL-Iin regard to the effect on carboplatin-induced mortality; doses of CA that failed to block carboplatin-induced mortality did not block IL-lreversal of carboplatin-induced mortality (Fig 2 ) . We hypothesize that ( 1 ) IL-1 induces production of inflammatory mediators (eg, TNF, interferons, a , ,& and 7 , macrophage inflammatory proteins, prostaglandins, and others) that inhibit hematopoiesis, and (2) that IL-I induces production of hematopoietic growth factors when given at nontoxic doses (0.5 to 2 pgldmouse). We further hypothe- 0 + UMMS 0 0.2 0.1 X lo5 0.3 0.4 0.5 CA Dose (mg/mouse/day) No IL-l 18, l ~ ~ 1X 2io3 ~ -e WBC I 1 0 3 0 04 0 0.1 0.2 0.3 0.4 I 0.5 CA Dose (mglmouselday) IL-l 8pg/mouse/day Fig 4. In two separateexperiments, 6-week-old C3HlHe.J mica were given IL-l (IP at 8 pglmouse), CA (SO at 0.1, 0.25, 0.5 mgl mouse), or both IL-l and CA for 5 days. Mice surviving were bled on day 6 by cardiac puncture for determination of peripheral blood counts illustrated in Fig 4 and plasma parametars illustrated in Fig 5. The number of animals surviving to day 6 and plasma TNF levels are shown in Table 3. From www.bloodjournal.org by guest on November 20, 2014. For personal use only. 1462 RINEHART ET AL t Glucose (mgldl 5 0 r "t BUN (mgldl) t SGOT (UIL) & Cholesterol (mgldl) ~ Iron (vgldl) i -b 0.1 0.2 0.3 0.4 1 0.5 CA Dose (mg/mouse/day) No IL-l Glucose (mg/dl BUN (mg/dl) t SGOT(UL) + Cholesterol (mg/dl) ++ Iron (pg/dl) "+ t B -6 0.1 0.2 0.3 0.4 0.5 CA Dose (mglmouselday) IL-l 8CcglmouseIday Fig 5. We have also observed that at high doses, CA (0.5 mg/ dlmouse) blocked carboplatin-induced death in our murine model. We chose carboplatin for use in these atudies because its only observed toxicity at the doses used are hematologic; mice died at the nadir of their peripheral blood counts and bacterial peritonitis was present at death. Furthermore, biochemical profiles obtained 24 hours before death in mice receiving carboplatin alone showed no decrease in renal or hepatic function (data not presented). We have n o w shown that high-dose cortisone acetate does ameliorate the hematopoietic toxicity of carboplatin."' It is likely that the mechanisms by which CA and IL-1 protect mice from the hematopoietictoxicities of carboplatinaredifferentbecause IL-l increases the number of hematopoietic precursors in spleen (CFU-GM), whereas at high doses, CA reduces hematopoieticprecursors. CA may act by increasing the number of hematopoietic precursors in G , or induction of other protective mechanisms (eg, induction of increased levels of glutathione or metalothiones). IL- 1 may providehematopoietic protection by increasing the total number of hematopoietic precursors. These observations may have clinicalutility. It may be possible to reduce toxicity of IL- I , IL-3, IL-6, or GM-CSF by concomitant administration of corticosteroids. We have shown in vitrothathydrocortisonesuccinate,prednisone, and dexamethasone enhanceIL- I , IL-3, and GM-CSF induction of human CFU-GM proliferationatconcentrations readily obtainable clinically with modest dosesof these corticosteroids."."." Furthermore, we have shown that GM-CSF andIL-l inducehuman mononuclear release of TNF and this release is blockedby the same concentrations of corticosteroids that enhance CFU-GM induction."' These findings suggest that a clinical trial to examine thecombination corticosteroids and IL-l or IL-3 may be feasible. See legend to Fig 4. ACKNOWLEDGMENT size that corticosteroids block production of these inflammatory inhibitors, but not hematopoietic growth factors. Data have been developed to support the first part of our hypothesis,ie, that IL-I administration induces production of inflammatory mediators such as TNF,21.2"2sand hematopoietic growth factors such as GM-CSF and IL-6.2h-2X The second part of our hypothesis, ie, thatcorticosteroids enhance production of hematopoietic growth factors and inhibit production of inflammatory mediators, is not supported by an observation suggesting that corticosteroidsinvitroinhibit GM-CSF p r o d ~ c t i o n . ~ ~ have W e recently examined thepharmacokinetics of CA in C3H/HeJ mice: CA administered SQ at 0.5 mg induces a cortisol (the active metabolite of CA) peak level of 50 pg/dL. This concentrationof hydrocortisone enhances IL-l-induced CFU-GM formation in vitro." We hypothesize that the concentrationsof corticosteroids needed to inhibit IL-l-induced hematopoietic growth factor release may be greater than those required to inhibit release of inflammatory cytokines. In addition, we are currently examining theeffect of corticosteroidson hematopoietic growth factor and inflammatory mediator mRNA expression in vivo and in vitro. We would like to acknowledge the support of William Buhles. PhD, DVM (Syntex Discovery Research) and Edward Rappaport, MD (Scott & White), in the development of these data. REFERENCES 1. Pettengell R, Gurney H, RadfordJA, Deakin DP,James R, Wilkinson PM, Kane K.Bentley J, Crowther D: Granulocyte colonystimulating factor to prevent dose-limiting neutropenia in non-HodgBlood 80:1430, kin'slymphoma: A randomizedcontrolledtrial. 1992 2. Advani R, Chao NJ, Homing SJ. Blume KG, Ahn DK. Lamhorn KR, Fleming NC, Bonnem EM, Greenberg PL: Granulocyteto macrophage colony-stimulating factor (GM-CSF) as an adjunct autologous hemopoieticstem cell transplantation for lymphoma. Ann Intern Med 116: 183, 1992 3. Neidhart JA, Mangalik A. Stidley CA, Tebich SL. Sarmiento LE, Ptile JE, Oette DH, Oldham FB: Dosing regimen ofgranulocytemacrophage colony-stimulating factorto support dose-intensive chemotherapy. J Clin Oncol 10: 1460, 1992 4. Neidhart JA, Mangalik A. Kohler W, Stidley C, Saiki J, Duncan P, Souza L, Downing M: Granulocyte colony-stimulating factor stimulates recovery of granulocytes in patients receiving dose-intensive chemotherapy without BM transplantation. J Clin Oncol 7: 1685. I989 From www.bloodjournal.org by guest on November 20, 2014. For personal use only. INTERACTIVEEFFECTS OF IL-l AND CORTICOSTEROIDS 5. Rinehart J: Phase 1 trial of IL-3, Carboplatin, and etoposide in patients with solid tumors. Blood 80:86a, 1992 (abstr, suppl) 6. Samuels B, Bukowski R, Gordon M, Samuel S, Isaac R, Demetri G: Phase 1 study of rhIL-6 with chemotherapy (CT) in advanced sarcoma. Proc Am Soc Clin Oncol 12:291, 1993 7. Olencki T, Budd GT, Murthy S, Finke J, Herzog P, McLain D, Tubbs R, Tuason M, Edinger M, Levitt D, Bukowski RM: Immunoregulatory and hematopoietic effects of interleukin-6 (rhIL-6) in cancer patients. Proc Am SW Clin Oncol 12:292, 1993 8. Smith J W , Longo DL, Alvord WG, Janik JE, Sharfman WH, Gause BL, Curti BD, Creekmore SP, Holmlund JT, Fenton RC: The effects of treatment with interleukin-la on platelet recovery after high-dose Carboplatin. N Engl J Med 328:756, 1993 9. Du XX, Nebem T, Goldman S, Williams DA: Effects of recombinant human interleukin-] 1 on hematopoietic reconstitution in transplant mice: Acceleration of recovery of peripheral blood neutrophils and platelets. Blood 81:27, 1993 10. Brandt J, Briddell RA, Srour EF, Leemhuis TB, Hoffman R: Role of c-kitligand in the expansion of human hematopoietic progenitor cells. Blood 79:634, 1992 11. Molineaux G, Migdalska A, Szmitkowski M, Zsebo K, Dexter TM: The effects of hematopoiesis of recombinant stem cell factor (ligand for c-kit) administered in vivo to mice either alone or in combination with granulocyte colony-stimulating factor. Blood 78:961, 1991 12. Hersh EM, Muggia FM, Whitehead RP, Rinehart JJ, Bonnet JD: Phase I1 studies of recombinant human tumor necrosis factor alpha (rHuTNFa) in patients with malignant disease. A summary of the Southwest Oncology Group experience. J Immunother 10:426, 1991 13. Creagan ET, Kovach JS, Moertel CG, Frytak S, Kvols LK: A phase I clinical trial of recombinant human tumor necrosis factor. Cancer 62:2467, 1988 14. Eliason JF and Vassalli P: Inhibition of hemopoiesis in murine marrow cell cultures by recombinant murine tumor necrosis factor Q: Evidence for long-term effects on stromal cells. Blood Cells 14:339, 1988 15. Hino M, Tojo A, Miyazono K, Urabe A, Takaku F: Effects of type p transforming growth factor on haematopoietic progenitor cells. Br J Haematol 70:143, 1988 16. Delamater L, Rinehart J: The effect of corticosteroids on IL-l induction of hematopoietic progenitor proliferation. Blood 80:413a, 1992 (abstr, suppl) 17. Rinehart JJ, Sagone A, Balcerzak SP, LoBuglio AF: Effect of corticosteroids on human monocyte function. J Clin Invest 54:1337, 1974 1463 18. Rinehart JJ, Sagone A, Balcerzak SP, Ackerman A, LoBuglio AF: Effect of corticosteroid therapy on human monocyte function. N Engl J Med 292:769, 1976 19. Boumpas DT, Paliogianni F, Anastassiou ED, Balow JE: Glucocorticosteroid action on the immune system: Molecular and cellular aspects. Clin Exp Rheumatol 9:413, 1991 20. Bailey JM: New mechanisms for effects of anti-inflammatory glucocorticoids. Refractors 3:97, 1991 21. Neta N, Oppenheim J: IL-l: Can we exploit Jekyll and subjugate Hyde? Biol Ther Cancer Updates 1O:l. 1992 22. Rajagopalan V, Essex D, Shapiro SS, Konkle BA: TNF-a modulation of glycoprotein I@ Q expression in human endothelial and erythroleukemia cells. Blood 80:153, 1992 23. Shalaby MR. Waage A, Aarden L, Espevik T Endotoxin, tumor necrosis factor Q and interleukin 1 induce interleukin 6 production in vivo. Clin Immunol Immunopathol 53:488, 1989 24. Dinarello CA: Interleukin 1 and its biologically related cytokines. Adv Immunol 44:153, 1989 25. Libert C, Brouckaert P, Shaw A, Fiers W: Induction of interleukin 6 by human and murine recombinant interleukin 1 in mice. Eur J Immunol 20:691, 1990 26. Bagby GC Jr, Dinarello CA, Wallance P, Wagner C, Hefeneider S, McCall E: Interleukin 1 stimulates granulocyte macrophage colony-stimulating activity release by vascular endothelial cells. J Clin Invest 78:1316, 1986 27. Fibbe WE, Van Damme J, Billiau A, Duinkerken N, Lurvink E, Ralph P, Altrock BW, Kaushansky K, Willemze R, Falkenburg JHF: Human fibroblasts produce granulocyte-CSF, macrophageCSF, and granulocyte-macrophage-CSFfollowing stimulation by interleukin-l and poly(rI).poly(rC). Blood 72:860,1988 28. Neta R, Vogel SN, Plocinski JM, Tare NS, Benjamin W, Chizzonite R, Pilcher M: In vivo modulation with anti-interleukin1 (&l) receptor (p80) antibody 35F5 of the response to 1L-l. The relationship of radioprotection, colony-stimulating factor, and IL-6. Blood 76:37, 1990 29. Thorens B, Mermod JJ, Vassalli P: Phagocytosis and inflammatory stimuli induced GM-CSF mRNA in macrophages through posttranscriptional regulation. Cell 48:671, 1987 30. Rinehart J, Delamater E, Keville L: Corticosteroids reduce hematopoietic toxicity of dose intensive chemotherapy in a murine model. Blood 82:652a, 1993 (abstr, suppl) 31. DiSanto AR, DeSante KA: Bioavailability and pharmacokinetics of prednisone in humans. J Pharm Sci 64:109, 1975 32. Ferry JJ, Horvath AM,Bekersky I, Heath EC, RyanCF, Colburn WA: Relative and absolute bioavailability of prednisone and prednisolone after separate oral and intravenous doses. J Clin Pharrnacol 28:81. 1988
© Copyright 2026