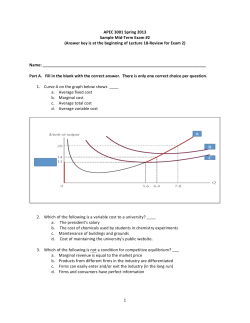
Equilibrium Worksheet (VC 3 & 4)
Chapter 15 Lesson 5 Worksheet Short Answer - answer the following questions. 1) The diagrams represent equilibrium mixtures for the reaction: A + X AX2. Is the reaction exothermic or endothermic? Explain. 2) Critique the following statement: When more silver chloride is added to the following reaction at equilibrium, AgCl (s) Ag+ (aq) + Cl- (aq), the system responds by forming more silver and chloride ions. 3) Given the following information: 2NO (g) NO (g) + Cl2 (g) H = 75kJ. What change occurs in the system at equilibrium a. If the temperature is decreased? b. If the partial pressure of chlorine gas is increased? c. If the total volume is increased? 4) An equilibrium involving N2O4 is established at 25°C in a 1.00 L vessel. N2O4 (g) 2NO2 (g) Kc = 4.61 x 10-3 at 25°C. The initial concentrations are 0.0500 mol NO2 and 0.100 mol N2O4. This vessel is connected to another vessel which has a volume of 3.00 L. If the system at equilibrium is permitted to expand into the 3.00 L vessel a. In what direction does the reaction proceed when equilibrium is established in the 1.00 L vessel? b. What direction will the reaction proceed in the larger 3.00 L volume? 5) For an exothermic reaction, increasing the reaction temperature results in a(an) __________ in the equilibrium constant, K. 6) If a reaction is endothermic, __________ the reaction temperature results in an increase in equilibrium constant, K. 7) For the Haber process reaction: N2 (g) + 3H2 (g) 2NH3 (g) a. If the [H2] is increased, i. Equilibrium will shift (left or right). ii. [N2] will (increase or decrease). iii. [NH3] will (increase or decrease). b. If the [N2] is decreased, i. Equilibrium will shift (left or right). ii. [H2] will (increase or decrease). iii. [NH3] will (increase or decrease). 8) In the equilibrium for this synthesis of methyl alcohol, CO (g) + 2H2 (g) CH3OH (g) + energy a. If the temperature is decreased, i. Equilibrium will shift (left or right). ii. [CH3OH] will (increase or decrease). b. If the [H2] is increased, i. Equilibrium will shift (left or right). ii. [CH3OH] will (increase or decrease). iii. The temperature in the vessel will (increase or decrease). 9) Consider this reaction at equilibrium. HCOO- (aq) + H2O (l) HCOOH (aq) + OH- (aq) a. If water is added, equilibrium will _____. b. If the [OH-] is increased, equilibrium will _____. i. [HCOOH] will (increase or decrease). ii. [HCOO-] will (increase or decrease). c. If H+ is added, equilibrium will _____. i. [HCOO-] will (increase or decrease). d. If a catalyst is added, equilibrium will _____. Multiple Choice – Choose the best answer possible. 10) The effect of a catalyst on an equilibrium is to __________. A) increase the rate of the forward reaction only B) increase the equilibrium constant so that products are favored C) slow the reverse reaction only D) increase the rate at which equilibrium is achieved without changing the composition of the equilibrium mixture E) shift the equilibrium to the right 11) Consider the following reaction at equilibrium.: 2CO2 (g) 2CO (g) + O2 (g) ΔH° = -514 kJ Le Châtelier's principle predicts that the equilibrium partial pressure of CO (g) can be maximized by carrying out the reaction __________. A) at high temperature and high pressure B) at high temperature and low pressure C) at low temperature and low pressure D) at low temperature and high pressure E) in the presence of solid carbon 12) Consider the following reaction at equilibrium: C (s) + H2O (g) CO (g) + H2 (g) Which of the following conditions will increase the partial pressure of CO? A) decreasing the partial pressure of H2O (g) B) removing H2O (g) from the system C) decreasing the volume of the reaction vessel D) decreasing the pressure in the reaction vessel E) increasing the amount of carbon in the system 13) Consider the following reaction at equilibrium: 2CO2 (g) 2CO (g) + O2 (g) Le Châtelier's principle predicts that an increase in temperature will __________. A) increase the partial pressure of O2 (g) B) decrease the partial pressure of CO2 (g) C) decrease the value of the equilibrium constant D) increase the value of the equilibrium constant E) increase the partial pressure of CO ΔH° = -514 kJ 14) Consider the following reaction at equilibrium: 2CO2 (g) 2CO (g) + O2 (g) ΔH° = -514 kJ Le Châtelier's principle predicts that adding O2 (g) to the reaction container will __________. A) increase the partial pressure of CO (g) at equilibrium B) decrease the partial pressure of CO2 (g) at equilibrium C) increase the value of the equilibrium constant D) increase the partial pressure of CO2 (g) at equilibrium E) decrease the value of the equilibrium constant 15) Consider the following reaction at equilibrium: 2NH3 (g) N2 (g) + 3H2 (g) Le Châtelier's principle predicts that the moles of H2 in the reaction container will increase with __________. A) some removal of NH3 from the reaction vessel (V and T constant) B) a decrease in the total pressure (T constant) C) addition of some N2 to the reaction vessel (V and T constant) D) a decrease in the total volume of the reaction vessel (T constant) E) an increase in total pressure by the addition of helium gas (V and T constant) True or False – Identify if the statement is true or false 16) The effect of a catalyst on a chemical reaction is to react with product, effectively removing it and shifting the equilibrium to the right. 17) At constant temperature, reducing the volume of a gaseous equilibrium mixture causes the reaction to shift in the direction that increases the number of moles of gas in the system. 18) In an exothermic equilibrium reaction, increasing the reaction temperature favors the formation of reactants. 19) Le Châtelier's principle states that if a system at equilibrium is disturbed, the equilibrium will shift to minimize the disturbance.
© Copyright 2026