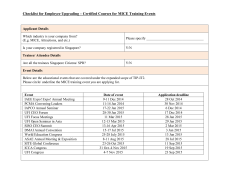
supplemental information - The Huguenard Lab
Neuron, Volume 78 Supplemental Information Endogenous Positive Allosteric Modulation of GABAA Receptors by Diazepam binding inhibitor Catherine A. Christian, Anne G. Herbert, Rebecca L. Holt, Kathy Peng, Kyla D. Sherwood, Susanne Pangratz-Fuehrer, Uwe Rudolph, and John R. Huguenard Inventory of Supplemental Information Figure S1. Effects of FLZ and the α3(H126R) Mutation on nRT sIPSC Duration Persist into Adulthood, Related to Figure 1. Figure S2. FLZ Preincubation Does Not Alter sIPSC Kinetics or Frequency in VB, Related to Figure 2. Figure S3. Map of AAV Genome Packaged Into AAV-DJ-CMV-DBI-T2A-hrGFP Virus, Related to Figure 5. Figure S4. Clonazepam Occludes nRT-Dependent Potentiation, and Combined GAT Blockade and FLZ Eliminates all nRT-Dependent Potentiation of Sniffer Responses, Related to Figure 6. Figure S5. α3(H126R) Mutants Exhibit More Severe Experimental Absence Seizures Than Wild-Types, Related to Figure 7. Table S1. Summary of nRT sIPSC parameter values (mean + SEM) for cells from WT and α3(H126R) mice with and without FLZ treatment. Table S2. Summary of nRT sIPSC parameter values (mean + SEM) for cells from WT and nm1054 mice. Experimental Procedures Supplemental References Christian et al. Supplemental Figures and Legends Figure S1. Effects of FLZ and the α3(H126R) Mutation on nRT sIPSC Duration Persist into Adulthood, Related to Figure 1. (A) sIPSC half width in individual wild-type nRT cells (n=9) in slices from P48-54 mice before (Con) and during FLZ treatment. Open boxes indicate mean + SEM during control and FLZ conditions. (B) Mean + SEM for sIPSC half width in WT (black bar, n=11 cells) and α3(H126R) mutant mice (blue bar, n=8 cells). *p<0.05 vs. WT; ***p<0.001 vs. Con. 2 Christian et al. Figure S2. FLZ Preincubation Does Not Alter sIPSC Kinetics or Frequency in VB, Related to Figure 2. (A-D) Probability distributions constructed from 100 randomly selected events per cell comparing half width (A), 90% width (B), weighted decay time constant (τd,w; C), and interevent interval (D) in VB cells under control (n=8, black lines) and FLZ-preincubated (n=4, red lines) conditions. 3 Christian et al. Figure S3. Map of AAV Genome Packaged Into AAV-DJ-CMV-DBI-T2A-hrGFP Virus, Related to Figure 5. Control AAV-DJ-CMV-hrGFP virus did not contain DBI and T2A sequences. Courtesy of Michael Lochrie, Stanford Neuroscience Gene Vector and Virus Core. 4 Christian et al. Figure S4. Clonazepam Occludes nRT-Dependent Potentiation, and Combined GAT Blockade and FLZ Eliminates all nRT-Dependent Potentiation of Sniffer Responses, Related to Figure 6. (A) Responses averaged across all WT patches in the presence of CZP, normalized to peak amplitude. (B) Mean + SEM for 90-10% decay time of responses (p>0.6) obtained in patches placed in VB (n=6) or nRT (n=6) in the presence of 100 nM – 1 µM CZP. (C) Responses averaged across all WT patches in the presence of GAT antagonists (4 µM NNC711 and 10 µM SNAP-5114), normalized to peak amplitude. (D) Responses averaged across all WT patches in the presence of GAT antagonists and FLZ, normalized to peak amplitude. (E) Mean + SEM for 90-10% decay time of responses obtained in the presence of GAT antagonists alone (turquoise bars, VB n=7, nRT n=6; p<0.001) or GAT antagonists and FLZ (magenta bars, VB n=6, nRT n=8; p>0.9). Patches placed in VB are not significantly different between the two groups (p>0.1). hν symbol – 1 ms UV laser stimulus. 5 Christian et al. Figure S5. α3(H126R) Mutants Exhibit More Severe Experimental Absence Seizures Than Wild-Types, Related to Figure 7. (A) Continuous EEG recordings showing 4-6 Hz activity in response to PTZ injection. (B) Mean + SEM for SWD rate following PTZ injection (Time 0) in WT (n=7) and α3(H126R) mice (n=4). Gray bars indicate times at which seizure parameters differed between mutants and controls (p<0.05). 6 Christian et al. Supplemental Tables Amplitude (pA) 1090% Rise time (ms) Half width (ms) 90% width (ms) τd,w (ms) sIPSC Charge (fC) τfast (ms) τslow (ms) % slow Freq. (Hz) WT control -20.58 + 1.11 1.31 + 0.05 122.14 + 6.24 255.61 + 16.63 105.74 + 3.05 -2033 + 134 39.59 + 2.15 177.02 + 11.50 65.14 + 3.09 1.02 + 0.18 WT+FLZ -25.64 + 2.84 ** vs. WT con 1.14 + 0.04 97.62 + 6.48 ***vs. WT con 229.93 + 16.81 97.91 + 5.78 *vs. WT con -2461 + 337 p=0.06 vs. WT con 29.49 + 2.89 **vs. WT con 152.09 + 9.95 *vs. WT con 64.26 + 3.64 1.15 + 0.32 α3(H126R) control -17.16 + 2.17 1.42 + 0.06 102.15 + 4.72 * vs. WT con 186.21 + 11.34 ** vs. WT con 93.70 + 4.66 * vs. WT con -1320 + 189 * vs. WT con 31.93 + 2.40 * vs. WT con 144.40 + 8.85 * vs. WT con 62.44 + 4.49 1.10 + 0.20 α3(H126R) +FLZ -18.69 + 1.94 p=0.069 vs. α3(H126R) con 1.35 + 0.06 107.24 + 9.72 181.09 + 16.30 91.42 + 7.10 -1548 + 169 p=0.06 vs. α3(H126R) con 41.38 + 4.19 158.70 + 19.82 49.94 + 4.22 1.48 + 0.36 Table S1. Summary of nRT sIPSC parameter values (mean + SEM) for cells from WT and α3(H126R) mice with and without FLZ treatment. Values calculated from P4-14 WT cells (n=23, 13 treated with FLZ) and α3(H126R) cells (n=20, 10 treated with FLZ). Comparisons between WT and α3(H126R) control groups performed using independent two-tailed t-tests. Effects of FLZ within each genotype assessed using paired t-tests. Note that 4 of 10 α3(H126R) cells exhibited an increase in sIPSC amplitude in response to FLZ, indicating that the increase in amplitude represents a non-specific effect, which is also reflected in the slight increase in charge transfer with FLZ treatment in both genotypes. Con, control; τd,w, weighted decay time constant. *p<0.05, **p<0.01, ***p<0.001. Related to Figures 1 and 2. 7 Christian et al. Amplitude (pA) 1090% Rise time (ms) Half width (ms) 90% width (ms) τd,w (ms) sIPSC Charge (fC) τfast (ms) τslow (ms) % slow Freq. (Hz) WT -11.78 + 0.27 1.53 + 0.04 71.19 + 3.09 98.27 + 4.23 55.48 + 1.80 -644.06 + 16.30 25.73 + 1.64 95.44 + 6.70 57.43 + 2.71 2.28 + 0.19 nm1054 -11.48 + 0.15 1.60 + 0.04 61.75 + 2.27 * 80.99 + 2.55 ** 49.74 + 1.85 * -566.51 + 16.64 ** 18.48 + 1.37 ** 74.98 + 5.28 * 64.77 + 3.72 3.43 + 0.28 ** Table S2. Summary of nRT sIPSC parameter values (mean + SEM) for cells from WT and nm1054 mice. Values calculated from P22-29 WT cells (n=14) and nm1054 cells (n=13). Comparisons between genotypes performed using independent two-tailed t-tests. Note that events are shorter than those in Supplementary Table 1 due to older age of animals. τd,w, weighted decay time constant. *p<0.05, **p <0.01 vs. WT. Related to Figure 5. 8 Christian et al. Extended Experimental Procedures Animals All procedures were approved by the Administrative Panel on Laboratory Animal Care at Stanford University. Mice were bred and housed on a 12:12 light-dark photoperiod with food and water available ad libitum. α3(H126R) mice: Wild-type (WT) C57BL/6 mice (Charles River Laboratories, Hollister, CA) were compared with α3(H126R) mice bred on a C57BL/6 background. Where indicated, some EEG experiments were performed using α3(H126R) mice generated on the 129X1/SvJ background (Löw et al., 2000) and compared to WT 129X1/SvJ mice (Jackson Laboratory, Bar Harbor, ME). The α3 subunit gene is located on the X-chromosome; mutant animals were thus either hemizygote male or homozygote female. nm1054 mice: Following an established breeding scheme (Ohgami et al., 2005), an initial cross of nm1054 heterozygote (Het) males with WT 129S6/SvEvTac female mice (Taconic Farms, Oxnard, CA) yielded Het male and female progeny, which were then bred to each other to produce WT, Het, and homozygous mutant littermates. Here we refer to the homozygous mutant mice as nm1054. No heterozygotes were used in this study. The 129S6/SvEvTac background was chosen because this mutation has been shown to demonstrate a high degree of prenatal and early postnatal lethality on the C57BL/6J background (Ohgami et al., 2005). 9 Christian et al. EEG recordings In this study we use “EEG” to refer to electrocorticography. For EEG apparatus implantation, mice older than P30 of either sex were anesthetized with ketamine (80 mg/kg i.p.) and xylazine (16 mg/kg i.p.). EEG recordings were obtained from either metal skull screws or silver wires implanted above the left and right frontal and parietal cerebral cortices. Animals were housed individually or in pairs following EEG apparatus implantation. At least 1 week later, mice were placed in the recording area and EEG activity and simultaneous video were recorded. Experimental absence seizures were induced in WT, α3(H126R), and nm1054 mutant mice via s.c. injection of pentylenetetrazol (PTZ, Tocris Bioscience, Ellisville, MO). EEG activity and simultaneous video were recorded for up to 90 min post-PTZ injection. Recordings in WT and α3(H126R) mutants on the 129X1/SvJ background strain, and in WT and nm1054 mice, used a 40 mg/kg dose of PTZ. In C57BL/6 mice, this dose was consistently observed to generate myoclonic jerks that often progressed to brief episodes of tonic-clonic convulsions (n=3 out of 4 mice, compared to 1 out of 12 129X1/SvJ mice tested), indicating a narrower therapeutic window for PTZ induction of SWDs in this strain. This is consistent with previous reports of induction of partial and generalized clonic seizures at this PTZ dose in C57BL/6 mice and related strains (Wong et al., 2003; Heurteaux et al., 2004; Hentschke et al., 2006; Cheung et al., 2010). Furthermore, SWD activity was disrupted in all animals following these convulsions. 10 Christian et al. Virus generation and injections The AAV-DJ serotype was chosen for its high degree of infection efficacy (Grimm et al., 2008; Xu et al., 2012). The AAV-DJ-CMV-DBI-T2A-hrGFP virus was generated in the Stanford Neuroscience Gene Vector and Virus Core (supported in part by NINDS grant P30 NS06937501A1) by calcium-phosphate-mediated transfection of AAV-293 cells (Agilent Technologies, Santa Clara, CA) and purified using an iodixanol step gradient (Hermens et al., 1999). For transfection 27 µg of pAAV CMV DBI-T2A-hrGFP vector, 27 µg of adenovirus helper plasmid (pHELPER, Agilent), and 27 µg AAV rep-cap helper plasmid (pRC-DJ, Mark Kay, Stanford) were used per T-225 flask of cells transfected. After ultracentrifugation, the iodixanol was diluted with TBS and the AAV was concentrated using 100 kDal molecular weight cutoff ultrafiltration devices (Millipore, Billerica, MA). The genomic titer (6.1 x 1012 vector genomes/ml) was determined by Q-PCR using primers that amplify and a probe that detects the hGH polyA region. Control AAV-DJ-CMV-hrGFP virus was also obtained from the Virus Core. The infectious titer was determined by infecting 3.0 x 105 HEK 293T cells on a 24-well plate in a volume of 0.3 ml in the presence of 4 µM etoposide. GFP+ cells were counted by fluorescence microscopy 2 days post-infection. Both viruses were injected at a concentration of 6.4 x 109 infectious units/ml. Bilateral stereotaxic injections of either control or DBI-expressing AAVs were performed under isoflurane anesthesia between P48-60. Injections were made using a 10-µl syringe, 34gauge needle, and a syringe pump (World Precision Instruments, Sarasota, FL) to control the injected volume (1 µl) and flow rate (120 nl/min). To target nRT, the stereotaxic coordinates were 1.25 mm posterior to Bregma, 1.9 mm lateral to the midline and 2.8 mm ventral to the dural surface. For injections in nm1054 mice, which typically have smaller brains (Lee et al., 2008), 11 Christian et al. the Bregma-Lambda distance was divided by 4.21 mm, the published standard distance (Paxinos and Franklin, 2001), and the coordinates were scaled accordingly for each mouse. Brain slices were prepared for electrophysiology as described below at 2-3 weeks post-injection. Infected cells expressing GFP were visualized using epifluorescence microscopy. No differences in recording properties within a group were observed between GFP-positive and –negative cells. Slices in which the injection targeting failed and fluorescence was only observed in VB were not included for experimentation. Brain slice preparation Mice of either sex at ages P4-81 were anesthetized with i.p. pentobarbital sodium (55 mg/kg) and killed via decapitation and the brain was quickly removed and placed in ice-cold (~4 o C) oxygenated (95% O2/5% CO2) sucrose slicing solution containing (in mM): 234 sucrose, 11 glucose, 26 NaHCO3, 2.5 KCl, 1.25 NaH2PO4, 10 MgSO4, and 0.5 CaCl2 (310 mOsm). Horizontal thalamic slices (250 µm thickness) containing nRT and VB were prepared as previously described (Huguenard and Prince, 1994) using a Leica VT1200 microtome (Leica Microsystems, Bannockburn, IL). Slices were incubated and continuously oxygenated in warm (~32oC) artificial cerebrospinal fluid (ACSF) containing (in mM): 10 glucose, 26 NaHCO3, 2.5 KCl, 1.25 NaHPO4, 1 MgSO4, 2 CaCl2, and 126 NaCl (298 mOsm) for 1 hour and then transferred to room temperature (~21-23 oC) for at least 15 min prior to recording. In some cases slices were preincubated by being placed in ACSF containing 100 nM clonazepam (CZP, Sigma, St. Louis, MO), 1 µM flumazenil (Hunkeler et al., 1981) (FLZ; also known as Ro15-1788, Sigma), the GAT-1 and GAT-3 antagonists 1,2,5,6-Tetrahydro-1-[2[[(diphenylmethylene)amino]oxy]ethyl]-3-pyridinecarboxylic acid hydrochloride (NNC-711, 4 12 Christian et al. µM, Tocris Bioscience, Minneapolis, MN) and 1-[2-[tris(4-methoxyphenyl)methoxy]ethyl]-(S)3-piperidinecarboxylic acid (SNAP-5114, 10 µM, Tocris) and/or 1 µM finasteride (FIN, Sigma) [45 min for FIN, a treatment duration demonstrated to remove endogenous neurosteroid effects (Tokuda et al., 2010)] when transferred to room temperature and maintained for >30 min prior to recording. Patch-clamp electrophysiology For recording, individual slices were placed in a recording chamber continuously superfused at 2 ml/min with oxygenated ACSF at room temperature. Slices were stabilized in the recording chamber >5 min before experimentation. Neurons in nRT or VB were visualized using a Zeiss Axioskop fixed-stage upright microscope (Carl Zeiss Inc., Thornwood, NY). Patchclamp recordings were made using a MultiClamp 700A amplifier with Clampex 9.2 software, and signals digitized using a Digidata 1322A (Molecular Devices, Sunnyvale, CA). Borosilicate glass recording pipettes were prepared using a Model P-97 Flaming/Brown micropipette puller (Sutter Instrument Co., Novato, CA) to 2-5 MΩ tip resistance when filled with intracellular pipette solution. Access resistance (Rs), measured from the peak of the averaged current response to 65 40-ms 5 mV depolarizing steps from a holding potential of -70 mV, was <20 MΩ in all whole-cell recordings. For within-cell drug treatments, data were discarded if access resistance increased >20% during recording. Appropriate vehicle controls (<0.2% DMSO) were included in the control ACSF solution. Only one cell per slice was recorded for within-cell drug treatment comparisons, no more than two cells from the same nucleus (nRT or VB) were recorded per slice, and no more than six cells in the same group were recorded per mouse. 13 Christian et al. IPSCs: Patch pipettes were filled with a near-isotonic chloride solution containing (in mM): 135 CsCl, 10 HEPES, 10 EGTA, 2 MgCl2, 5 QX-314, and pH adjusted to 7.3 with CsOH (290 mOsm). Recordings were not corrected for an estimated -5 mV liquid junction potential. To isolate GABAergic IPSCs, ionotropic glutamate receptors were blocked with either kynurenic acid (1 mM, Ascent Scientific, Princeton, NJ) or a combination of D-(-)-2-amino-5phosphonovaleric acid (APV, 100 µM, Ascent) plus 6,7-dinitroquinoxaline-2,3-dione (DNQX, 20 µM, Ascent). Cells were recorded in voltage-clamp mode with membrane potential clamped at -60 mV. For recordings of spontaneous IPSCs, signals were recorded in gap-free mode and low-pass filtered at 2 kHz or 4 kHz in nRT and VB, respectively, with gain set at 20 mV/pA. For evoked intra-nRT IPSCs, a bipolar tungsten stimulating electrode was placed in nRT ~100-200 µm from the recording electrode. Threshold intensity (10-40 V, 80-100 µs pulse width) was defined as that which produced 50% successes and 50% failures, and evoked currents were recorded at 1.5X threshold at 10 s stimulation intervals. GABA uncaging: Outside-out patches of membrane from nRT or VB cells were obtained by slowly pulling away the recording pipette shortly after breaking into the whole-cell configuration. Stable patches were maintained in voltage-clamp mode at a membrane holding potential of -30 mV. In all cases the recording pipette was pulled completely out of the slice to ensure total patch excision. Sniffer patches were placed ~25-50 µm into the slice, a depth at which cell bodies could be visualized easily. CNB-caged GABA (Invitrogen, Carlsbad, CA) was added to a recirculating 10-20 ml bath solution containing APV and DNQX. 0.1 mM caged GABA was 14 Christian et al. used for VB patch recordings, and 1 mM was used for nRT patch recordings, to account for the lower GABA affinity of nRT patches (Schofield and Huguenard, 2007). An ultraviolet laser beam (355 nm wavelength; DPSS Lasers, Santa Clara, CA) was directed into the epifluorescence port of the microscope and through the back aperture of a 60X water immersion objective. The tip of the recording pipette was positioned in the center of the laser spot to ensure maximal photolysis of caged GABA focused near the membrane patch. 1 ms-duration laser pulses were delivered at 10 s intervals. Recordings were low-pass filtered at 2 kHz with gain set at 20 mV/pA. For experiments in which patches pulled from VB were moved to nRT, final localization of the patch electrode tip in nRT was confirmed by capturing a video frame from the microscope using a 2.5X or 10X objective. Placement of patches in VB or nRT was alternated to ensure that conditions of slice incubation time and health were equivalent across groups. Histology and immunocytochemistry For DBI staining in Figures 5A-B, mice were anesthetized with Beuthanasia-D (110mg/kg) and perfused transcardially with saline followed by 4% paraformaldehyde (PFA, Sigma) in 0.1 M phosphate buffer at pH 7.4. The brains were removed and post-fixed in 4% PFA at 4oC overnight, then cryoprotected in 30% sucrose buffer and frozen on dry ice. Horizontal 50 µm slices were cut with a sliding microtome (Microm; HM 400). Free-floating sections were incubated for 1 hr in 10% Normal Donkey Serum followed by incubation with primary antibodies against DBI (rabbit polyclonal, 1:50; Santa Cruz Biotechnology, Santa Cruz, CA) at 4oC for 48 hrs. Sections were then rinsed in PBS and incubated for 2 hrs with corresponding fluorescent secondary antibodies (Jackson ImmunoResearch Laboratories, West Grove, PA). Sections were mounted on slides and coverslipped with Vectashield mounting media (Vector 15 Christian et al. Laboratories, Burlingame, CA). Z-stacks of images with an optical distance of 0.5 microns were captured with a laser scanning confocal microscope (Zeiss LSM 510) using 40X and 63X oilimmersion objectives. Secondary antibodies tagged to Fluorescein 488 and Cy3 were excited with 488 and 594 nm lasers and observed through 510-530 and 560-615 emission filters, respectively. A pinhole of 1 airy unit and identical settings for the detector gain and amplifier offset were used to capture all confocal images. Virus-infected slices were fixed in 4% PFA overnight after recording, washed in PBS, and resectioned to 50 µm thickness. Free-floating sections were then treated as described above using primary antibodies for DBI. The hrGFP signal produced following viral infection was strong enough to be imaged without the use of antibody amplification. Data analysis and statistics sIPSCs were analyzed using the custom software programs wDetecta and WinScanSelect (J.R.H., http://huguenardlab.stanford.edu/apps/wdetecta). Event detection threshold was confirmed for each cell and was typically set at 4-8 pA above baseline. sIPSCs were sorted into type-1 (those that decayed completely to baseline before the initiation of a subsequent event), type-2 (those whose decay was interrupted by a subsequent event), and type-3 (those whose initiation occurred during the decay phase of a previous event). The weighted decay time constant (τd,w) was calculated by dividing the total charge transfer (in fC) by the peak amplitude (in pA). To calculate τfast and τslow for a given cell, averaged type-1 events were obtained for each cell and fit with a double exponential function using Clampfit with DC offset set to 0. The decay of the averaged IPSC was fit to the following equation: I=A1e−t/τ1+A2e−t/τ2, in which τ1 represented τfast. The percentage of the decay represented by the slow component (% slow) was 16 Christian et al. calculated by the function A1/(A1+A2). For nonstationary variance analysis (Sigworth, 1980; De Koninck and Mody, 1994; Schofield and Huguenard, 2007), only type-1 events with rise times <2 ms were used. A mean current response was obtained by averaging all such events recorded in control conditions for each cell, and the mean current was normalized to the peak amplitude of each individual event. The variance from the cell mean trace was calculated for each individual sIPSC from the peak-to-baseline return portion of the event, and the mean variance and mean amplitude for each cell was divided into 100 equal bins. These data were plotted and fit to the parabolic function σ2-σ2Noise = iIm − I2m/N where σ2 is the variance, Im is the mean current, i is the unitary current, N is the number of channels, and σ2Noise is the variance in current baseline noise for the 30 ms preceding event onset. Evoked IPSC and uncaging recordings were analyzed using Clampfit. EEG recordings were analyzed using a continuous wavelet transform method in MATLAB (MathWorks, Natick, MA) to isolate SWD events (Schofield et al., 2009). Detection parameters were set to identify SWD events as periods in which the scale-averaged wavelet power was above a 99% confidence level threshold. Wavelet power ranges for positive detection were set in the 160 to 333 ms band (3 to 6.25 Hz) for screw recordings, or the 160 to 250 ms band (4 to 6.25 Hz) for wire recordings. Events with duration less than 800 ms were rejected and subsequent events separated by a gap less than 800 ms long were merged. Identification of spontaneous SWD events was confirmed via manual analysis of the power spectrum of each event in Clampfit 9.2 (Molecular Devices). Data were transferred to Excel (Microsoft, Redmond, WA), Origin 7 (Microcal Software, Northampton, MA), and SigmaStat (Aspire Software, Ashburn, VA) for statistical analysis. Comparisons between groups were made using two-tailed independent t-tests, nonparametric 17 Christian et al. Mann-Whitney Rank Sum Tests, or one-way ANOVA with Tukey’s post hoc means comparison tests. Within-cell comparisons were performed using two-tailed paired t-tests. Cumulative probability distributions were constructed using up to 100 randomly selected sIPSCs (events) per cell and compared using two-sample Kolmogorov-Smirnov (KS) goodness of fit tests. For amplitude and kinetics analyses, only type-1 events were used; all three types of events were used in analysis of frequency and interevent interval. Differences within each genotype for EEG parameters across different time points after PTZ injection were assessed using one-way repeated measures ANOVA. Data are presented as means + SEM. Statistical significance was set at p<0.05 for means comparisons, and p<0.001 for KS tests. Supplemental References Cheung, C.C., Yang, C., Berger, T., Zaugg, K., Reilly, P., Elia, A.J., Wakeham, A., You-Ten, A., Chang, N., Li, L., et al. (2010). Identification of BERP (brain-expressed RING finger protein) as a p53 target gene that modulates seizure susceptibility through interacting with GABAA receptors. Proc Natl Acad Sci USA 107, 11883–11888. Grimm, D., Lee, J.S., Wang, L., Desai, T., Akache, B., Storm, T.A., and Kay, M.A. (2008). In vitro and in vivo gene therapy vector evolution via multispecies interbreeding and retargeting of adeno-associated viruses. J Virol 82, 5887–5911. Hentschke, M., Wiemann, M., Hentschke, S., Kurth, I., Hermans-Borgmeyer, I., Seidenbecher, T., Jentsch, T.J., Gal, A., and Hübner, C.A. (2006). Mice with a targeted disruption of the Cl/HCO3- exchanger AE3 display a reduced seizure threshold. Mol Cell Biol 26, 182–191. Hermens, W.T., Ter Brake, O., Dijkhuizen, P.A., Sonnemans, M.A., Grimm, D., Kleinschmidt, J.A., and Verhaagen, J. (1999). Purification of recombinant adeno-associated virus by iodixanol gradient ultracentrifugation allows rapid and reproducible preparation of vector stocks for gene transfer in the nervous system. Hum Gene Ther 10, 1885–1891. Heurteaux, C., Guy, N., Laigle, C., Blondeau, N., Duprat, F., Mazzuca, M., Lang-Lazdunski, L., Widmann, C., Zanzouri, M., Romey, G., et al. (2004). TREK-1, a K+ channel involved in neuroprotection and general anesthesia. EMBO J 23, 2684–2695. Lee, L., Campagna, D.R., Pinkus, J.L., Mulhern, H., Wyatt, T.A., Sisson, J.H., Pavlik, J.A., Pinkus, G.S., and Fleming, M.D. (2008). Primary ciliary diskinesia in mice lacking the novel ciliary protein Pcdp1. Mol Cell Biol 28, 949–957. 18 Christian et al. Ohgami, R.S., Campagna, D.R., Antiochos, B., Wood, E.B., Sharp, J.J., Barker, J.E., and Fleming, M.D. (2005). nm1054: a spontaneous, recessive, hypochromic, microcytic anemia mutation in the mouse. Blood 106, 3625–3631. Paxinos, G., and Franklin, K.B.J. (2001). The Mouse Brain in Stereotaxic Coordinates (San Diego, CA: Academic Press). Wong, M., Wozniak, D.F., and Yamada, K.A. (2003). An animal model of generalized nonconvulsive status epilepticus: immediate characteristics and long-term effects. Exp Neurol 183, 87–99. Xu, W., Morishita, W., Buckmaster, P.S., Pang, Z.P., Malenka, R.C., and Südhof, T.C. (2012). Distinct neuronal coding schemes in memory revealed by selective erasure of fast synchronous synaptic transmission. Neuron 73, 990–1001. 19
© Copyright 2026