Vaccination versus treatment of influenza in working adults: a cost-effectiveness analysis
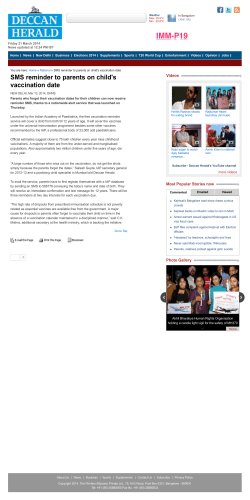
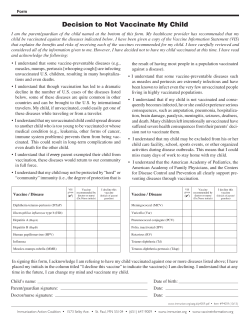
The American Journal of Medicine (2005) 118, 68-77 CLINICAL RESEARCH STUDY Vaccination versus treatment of influenza in working adults: a cost-effectiveness analysis Michael B. Rothberg, MD, MPH,a,b David N. Rose, MDa,b a From the Division of General Medicine and Geriatrics, Department of Medicine, Baystate Medical Center (MR), Springfield, Massachusetts, and b Tufts University School of Medicine (DNR), Boston, Massachusetts. KEYWORDS: Influenza; Vaccination; Antiviral therapy; Cost-effectiveness; Decision model Purpose: To determine the cost-effectiveness of influenza vaccination, antiviral therapy, or no intervention for healthy working adults, accounting for annual variation in vaccine efficacy. Methods: We conducted a cost-effectiveness analysis based on published clinical trials of influenza vaccine and antiviral drugs, incorporating 10 years of surveillance data from the World Health Organization. We modeled influenza vaccination, treatment of influenza-like illness with antiviral drugs, or both, as compared with no intervention, targeting healthy working adults under age 50 years in the general community or workplace. Outcomes included costs, illness days, and quality-adjusted days gained. Results: In the base case analysis, the majority of costs incurred for all strategies were related to lost productivity from influenza illness. The least expensive strategy varied from year to year. For the 10-year period, antiviral therapy without vaccination was associated with the lowest overall costs ($234 per person per year). Annual vaccination cost was $239 per person, and was associated with 0.0409 quality-adjusted days saved, for a marginal cost-effectiveness ratio of $113 per quality-adjusted day gained or $41,000 per quality-adjusted life-year saved compared with antiviral therapy. No intervention was the most expensive and least effective option. In sensitivity analyses, lower vaccination costs, higher annual probabilities of influenza, and higher numbers of workdays lost to influenza made vaccination more cost-effective than treatment. If vaccination cost was less than $16 or time lost from work exceeded 2.4 days per episode of influenza, then vaccination was cost saving compared with all other strategies. Conclusion: Influenza vaccination for healthy working adults is reasonable economically, and under certain circumstances is cost saving. Antiviral therapy is consistently cost saving. © 2005 Elsevier Inc. All rights reserved. Influenza affects 5% to 10% of the workforce annually, costing billions of dollars in lost productivity.1 Although influenza vaccination is cost saving in elderly and high-risk patients,2-5 results of cost-effectiveness studies involving Requests for reprints should be addressed to Michael Rothberg, MD, MPH, Division of General Medicine and Geriatrics, Baystate Medical Center, 759 Chestnut Street, Springfield, Massachusetts 01199, or E-mail address: [email protected]. 0002-9343/$ -see front matter © 2005 Elsevier Inc. All rights reserved. doi:10.1016/j.amjmed.2004.03.044 younger samples have been conflicting, even when accounting for the economic effect of time lost from work.3,6-10 Because hospitalizations or deaths due to influenza are rare among healthy adults,11 vaccination mostly decreases morbidity. Annual vaccination could potentially prevent most cases of influenza. However, 10 to 40 workers must be vaccinated to prevent 1 case.12 Giving antiviral drugs to persons who develop influenza-like illness is an alternative approach. Although antiviral therapy may work for more than half of all recipients, it Rothberg and Rose Vaccination versus Treatment of Influenza Annual Vaccination Antiviral Therapy No Intervention } Healthy Immune to A M Immune to B Immune A & B } 69 No ILI Treat Physician Visit Test and Treat + ILI No Intervention No Physician Visit } No Influenza Influenza A Influenza Influenza B } No Complication Hospitalization Emergency Room Visit Complication Home Antibiotics Figure 1 Markov model of yearly influenza infection. The decision to vaccinate is made before the start of the influenza season, at which time all cohort members begin in the healthy state. Each week, subjects may develop influenza-like illness, which may be followed by a physician visit. The physician decides, based on the current incidence of influenza, whether to treat, test, or withhold treatment (see Methods for details). Patients with influenza may develop complications, including hospitalization, emergency department visit, or bacterial infection requiring antibiotics. At the end of each week, cohort members return to the appropriate healthy or immune state. At the end of the season, all members return to the healthy state. Squares denote decision nodes; M denotes weekly Markov cycle; circles denote chance events; diamonds denote return to next Markov cycle; A and B refer to influenza types. ILI ⫽ influenza-like illness. shortens the course of but does not prevent the illness, and it is more expensive per course than vaccination. Rapid diagnostic testing can improve the likelihood that only patients with influenza will receive treatment, but testing is also expensive. Randomized controlled trials conducted in a single year or location are limited by annual variations in influenza incidence and vaccination efficacy, and their results are therefore not generalizable.6 We constructed a decision analysis model to determine the cost-effectiveness of influenza vaccination, antiviral therapy, or both for healthy working adults. To make sense of discordant trial results, we incorporated yearly variations in influenza rates and vaccine efficacy from 10 influenza seasons. We also examined the effect of worksite-specific variables, such as vaccination costs, the value of a workday, and the number of workdays lost to influenza. Methods Decision analysis model We constructed a Markov model using Decision Maker 7.07 (Pratt Medical Group, Boston, Massachusetts) to compare the outcomes of four strategies for healthy working adults under 50 years of age: annual vaccination; two antiviral therapy strategies for patients presenting with influenza-like illness; and no intervention. To model vaccination and treatment efficacy, we used World Health Organization (WHO) and Centers for Disease Control and Prevention (CDC) reports of the week-by-week incidence of influenzalike illness, and confirmed influenza infections by type from 1993 through 2002 (Appendix). The outcomes calculated were cost and quality-adjusted days gained. We made the following assumptions. The decision to vaccinate or to treat with antiviral therapy is made at the beginning of the influenza season. Vaccination is provided in a low-cost setting, such as an employee health service, and antiviral therapy is prescribed in a physician’s office. Influenza-like illness is defined as an abrupt onset of fever plus cough, headache, sore throat, or malaise. Antiviral therapy is initiated only if the patient presents within 48 hours of symptom onset, and only when influenza infections are reported in the previous week to comprise 10% or greater of cases of influenza-like illness. The CDC reports this information on its website14 and elsewhere. The choice of antiviral medication is made when the patient presents to the physician. We modeled two antiviral drug strategies that have been shown to be reasonable when influenza prevalence is under 30%: empirical amantadine and rapid testing followed by oseltamivir if results are positive.15,16 Both drugs are effective against influenza type A and relatively safe. Oseltamivir is more expensive, but is effective against influenza type B. The model begins with all patients in the healthy state at the start of the influenza season (Figure 1). During each weekly cycle, patients may contract influenza-like illness caused by influenza A, influenza B, or other viruses. Those not contracting the illness remain in the healthy state. Ill 70 The American Journal of Medicine, Vol 118, No 1, January 2005 Table 1 Values, utilities, and costs used in the analysis, and ranges tested in sensitivity analyses Variable Baseline* Influenza-like illness (per week) Influenza, given influenza-like illness Influenza B, given influenza Physician office visit, given influenza-like illness Complication of influenza requiring antibiotics Hospitalization, given influenza Average work loss from influenza (days) Length of hospitalization (days) Vaccination Efficacy against matched strain Side effects Minor Guillain-Barré Syndrome Influenza rapid test Sensitivity Specificity Antiviral therapy Shortens duration of illness by (hours) Amantadine Oseltamivir Efficacy against complications requiring antibiotics Amantadine Oseltamivir Side effects Amantadine Oseltamivir Utilities‡ Influenza Hospitalization Antiviral drug side effects Vaccine minor side effects Costs ($) Vaccine§ Vaccine administration Drugs, per course§ Amantadine Oseltamivir Antibiotics for influenza complications Rapid test Moderate complexity office visit Emergency department visit Hospitalization for influenza Guillain-Barré Syndrome Workday 0.02 Varies by week and year Varies by week and year 0.4 0.10 0.004 1.9 4.4 Range* Reference 0.005–0.03 0–0.31 0–1 0–1 0.05–0.17 0.001–0.007 0.5–5.0 3.9–4.8 14 14 14 6,17 18–20 21–23 8,24,25 26 0.72 0.54–0.83 12 0.64 1 ⫻ 10⫺6 0.10–0.70 1 ⫻ 10⫺5–1 ⫻ 10⫺7 27 28 0.65 0.99 0.50–0.78 0.95–1.00 Manufacturer† Manufacturer† 17.5–31.0 24–72 29 18 0 0.53 0 0.08–0.76 No data 18,20 0.09 0.10 0–0.26 0.075–0.11 29–32 18,20,33,34 0.6 0.4 0.88 0.999 0–1 0–0.5 0.5–1.0 0.8–1.0 9,10 9,10 Estimate Estimate 8.07 1.80 4.50–8.25 1.80–3.60 35 36 24 24 1.57 59.54 41.84 14.5 46 160 3663 124,737 177 0–25 3663–4957 0–500 37 37 37 List price 38 26 26,39 40 41 *Values are probabilities unless otherwise stated. †Zstatflu-ZymeTx, Oklahoma City, Oklahoma. ‡Range from 1.0 (perfect health) to 0 (death). §Component cost of the vaccine or drug. The total cost of treatment also includes the value of 30 minutes of worker’s time. patients may consult a physician, who may prescribe empiric therapy or perform rapid testing. Patients with influenza may develop complications requiring antibiotics, emergency department visits, or hospitalization. After recovering from influenza, patients are immune to the corresponding influenza type for the remainder of that season. At the end of the season, all patients return to the healthy state and the process is repeated for the next season. We calculated outcomes with base case parameter values and then performed sensitivity analyses using a wide range of published values (Table 1). We assumed that workers miss an average of 1.9 days for an influenza-like illness, based on three U.S. studies,8,24,25 but tested the range from 0.5 to 5.0 days in the sensitivity analysis. Because work loss occurs only on weekdays, weekend days count toward quality-adjusted days but do not accrue indirect costs from work loss. We assumed that 40% of patients with influenza-like illness visit a physician6,8,17,24 and that all visits take place within 48 hours of symptom onset. Antibiotic-requiring complication rates were based on the experience of subjects in the placebo arms of treatment trials.18,19 Hospitalization rates were based on rates calcu- Rothberg and Rose Vaccination versus Treatment of Influenza lated for healthy young women21 and in healthy patients under 65 years of age,22,23 which correlate closely with the hospitalization rate among placebo recipients in influenza treatment trials.42 Because influenza deaths in the young are rare and there are no available data on mortality rates, we assumed that no patients die of influenza. Vaccine efficacy varies from year to year, depending on how well the vaccine matches the circulating strains. In well-matched years, efficacy against serologically proven influenza is 72%.12 For each simulated year, we calculated vaccine efficacy by multiplying 0.72 by the percentage of circulating viruses that matched the vaccine strain in that year.13,43,44 Adverse effects of influenza vaccine include local soreness at the injection site,27 and, rarely, GuillainBarré syndrome.28 We estimated that adverse effects other than Guillain-Barré syndrome last for 2 days. The efficacy and side effects of antiviral drugs were taken from randomized placebo-controlled trials of average-risk subjects with naturally occurring infection. Antiviral drugs decreased symptoms by 1 to 1.5 days,18 and none have been proven to be more effective than another. We modeled amantadine because it is the least expensive in its class and oseltamivir because it is the only oral neuraminidase inhibitor. Only neuraminidase inhibitors have been shown to prevent bacterial complications of influenza.18,20 Both drugs have a similar incidence of side effects.18,20,29-34 Utilities are used to measure the quality of life in various health states, using a scale from 1.0 for perfect health to 0 for death. Previous cost-effectiveness models of influenza vaccine, which employed the Quality of Well-Being Index, assigned influenza illness a value of 0.6.9,10 Although some studies suggest that this estimate may be too high,45 we used it to ease comparison between studies. In the sensitivity analysis, we tested the full range from 0 to 1. Costs We took a societal perspective in keeping with the recommendations of the Panel on Cost-Effectiveness in Health and Medicine.46 Thus, we considered direct medical costs, as well as the indirect cost of lost productivity due to illness, using the average hourly cost of compensation for all civilian workers.41 Physician fees were based on a moderatecomplexity office visit for an established patient.38 We used the retail price provided by the manufacturer for the rapid diagnostic test (Zstatflu-ZymeTx, Oklahoma City, Oklahoma). Vaccination costs included the cost of the vaccine, 30 minutes of a worker’s time, and 5 minutes of a nurse’s time. Medication costs were average wholesale prices plus 30 minutes of a worker’s time to fill the prescription. Hospitalization costs were based on 333 patients hospitalized for influenza at 75 hospitals.26 All costs are converted to 2001 dollars using the medical care component of the Consumer Price Index.47 71 Sensitivity analysis We performed one-way and multiway sensitivity analyses to determine if reasonable variations in parameter values substantially affect the decision. We also performed a probabilistic (Monte Carlo) analysis to determine the probability that each intervention is reasonably cost-effective. We used normal distributions (for variables with values ⬎1) and logit distributions (for values between 0 and 1) based on the 95% confidence intervals reported in the literature. We then performed 1000 analyses, each calculated with randomly chosen values from these variable distributions. Results For the overall 10-year period, amantadine therapy was the least expensive strategy, costing $234 per person per year, and resulting in 0.0102 quality-adjusted days gained per person per year as compared with no intervention (Table 2). Annual vaccination cost was $4.64 more than amantadine therapy and resulted in 0.0409 quality-adjusted days gained, for a marginal cost-effectiveness ratio of $113 per qualityadjusted day gained or $41,000 per quality-adjusted lifeyear (QALY) saved. Oseltamivir therapy was less effective than annual vaccination and cost more per quality-adjusted day gained. For patients who were unwilling to take amantadine, annual vaccination had a cost-effectiveness ratio of $46 per quality-adjusted day or $17,000 per QALY saved. The least expensive strategy changed from year to year, as a result of variations in circulating influenza strains and the match between the strains and the vaccine (Figure 2). For every influenza season except 1997–1998, annual vaccination was the most effective strategy, and in four of 10 seasons it was also the least expensive. In half the years it was less expensive than no intervention, which was always the least effective strategy. In a hypothetical cohort of 1000 healthy working adults, amantadine therapy had no effect, as compared with no intervention, on the number of cases of illness, hospitalizations, or physician office visits, but reduced the number of lost workdays by 16% (Table 3). Annual vaccination had a greater effect than did no intervention, reducing by 57% the numbers of cases of influenza, lost workdays, and hospitalizations. However, annual vaccination reduced cases of influenza-like illness and physician office visits by only 8%. Total costs for the cohort ranged from $234,000 for amantadine therapy to $239,000 for annual vaccination, with the majority of costs due to lost productivity from influenza-like illness (Table 4). Amantadine saved $3000 and vaccination saved $6000 in lost productivity compared with no intervention. These savings were partially offset by the cost of amantadine and wholly offset by the cost of vaccination. 72 The American Journal of Medicine, Vol 118, No 1, January 2005 Table 2 Results of base case analysis of four influenza strategies for healthy working adults Strategy Amantadine therapy No intervention Rapid test-oseltamivir therapy Annual vaccination Cost ($) Per Year Effectiveness (Illness Days Per Year) Incremental Cost ($) Incremental Effectiveness (Illness Days Avoided) 234 236 237⫺ 239 1.75 1.76 1.73 1.63 — 2.31 2.51 4.64 — ⫺0.006 0.018 0.124 Quality-Adjusted Days Lost Amantadine therapy No intervention Rapid test-oseltamivir therapy Annual vaccination 234 236 237 239 Incremental Costeffectiveness Ratio ($/Illness Day Avoided) — Dominated* Extended dominance† 37 Quality-Adjusted Days Gained 0.692 0.702 0.692 0.651 — 2.31 2.51 4.64 — ⫺0.0102 0.0002 0.0409 Dominated* Extended dominance† 113 *Removed because strategy costs more and is less effective than amantadine therapy. †Removed because strategy is less cost-effective than vaccination. Sensitivity analysis Worksite-specific variables Assuming an average rate for influenza-like illness of 2% per week during influenza season, the model predicted an annual influenza infection rate that varied from 6% to 10% during the 10-year study period (Table 5). For the 3 years when the rate fell below 8%, annual vaccination was expensive as compared with amantadine therapy ($222 per quality-adjusted day saved to $682 per quality-adjusted day saved). In 4 of the years when the rate exceeded 8%, annual vaccination was cost saving. In assessment of the least expensive strategy based on three worksite-specific factors (the number of workdays lost per episode of influenza, the value of those days, and the cost of vaccination), annual vaccination was more cost-effective for workers who missed more days of work because of influenza illness (Figure 3). Beyond 2.4 days of work lost per episode of influenza illness, annual vaccination saved money relative to the amantadine therapy strategy. In contrast, if workers typically lost 1 day of work, annual vaccination still improved health, but at a 0.08 20 0.06 Cost ($) 0.02 0 0 Cost ($) Quality-Adjusted Days Gained 0.04 10 -0.02 Quality-adjusted days gained -10 -0.04 19921993 19931994 19941995 19951996 19961997 19971998 19981999 19992000 20002001 20012002 Influenza Season Figure 2 Cost and effectiveness of annual vaccination as compared with no intervention over 10 consecutive influenza seasons. The dark bars represent the difference in cost between the two strategies; the white bars represent the difference in quality-adjusted life-years (QALYs). Positive numbers mean that annual vaccination increases costs or QALYs as compared with no intervention. Rothberg and Rose Vaccination versus Treatment of Influenza 73 Table 3 Annual effect (effectiveness) of four influenza vaccination and treatment strategies on a cohort of 1000 healthy adult workers, averaged over 10 years Strategy Cases of Influenza-Like Illness Cases of Influenza Lost Workdays from Influenza Hospitalizations Courses of Antiviral Therapy Rapid Office Tests Performed Physician Office Visits 0 0 264 (255–279) 0 264 (255–279) Number (Range) No intervention Amantadine therapy Rapid testoseltamivir Annual vaccination 659 (636–698) 86 (65–103) 164 (123–201) 0.34 (0.30–0.40) 659 (636–698) 86 (65–103) 139 (107–169) 0.34 (0.30–0.40) 158 (131–199) 659 (636–698) 86 (65–103) 144 (109–175) 0.34 (0.30–0.40) 21 (15–27) 610 (581–645) 37 (27–72) 71 (51–138) 0.15 (0.10–0.30) marginal cost of $753 per quality-adjusted day saved as compared with amantadine therapy. For nonworking adults, no intervention was the least expensive strategy. Amantadine therapy cost $24 per quality-adjusted day saved compared with no intervention; annual vaccination cost $186 per quality-adjusted day saved compared with amantadine therapy. Changes as small as $5 in the overall cost of vaccination made annual vaccination either cost saving or more than $233 per quality-adjusted day gained. The cutoff of $5 represents just over 13 minutes of a worker’s time, and formulations of the vaccine vary in price by as much as $3.50 per dose. Monte Carlo analysis No intervention was never favored under any circumstances (Figure 4). Annual vaccination was always the most effective strategy, as well as the least expensive strategy 18% of the time. Amantadine therapy was the least expensive the rest of the time. Annual vaccination cost less than $100 per quality-adjusted day gained 40% of the time, and less than $300 per quality-adjusted day gained 77% of the time. Rapid testing followed by oseltamivir was more costeffective than the other two strategies less than 4% of the time. 0 158 (131–199) 264 (255–279) 0 244 (229–257) Discussion Our analysis of influenza strategies for working adults accounted for variations in the match between circulating influenza strains and vaccine components over a 10-year period, and tested both annual vaccination and the option of treating persons who develop influenza symptoms. During this period, antiviral therapy with amantadine, but not oseltamivir, was found to be consistently cost saving compared with no intervention, and overall was the least expensive strategy, although it provided fewer health benefits than did annual vaccination. Both the benefits and costs associated with vaccination were small. On average, vaccination could be expected to prevent 49 cases of influenza per 1000 workers vaccinated, avoiding 93 lost workdays, or 0.09 days per employee, at a net cost of approximately $3 per employee vaccinated. We also found that the determinants of cost-effectiveness fell into two categories: worksite-specific and influenza season–specific. Worksite variables, such as vaccination cost and the average number of sick days taken by a worker because of influenza infection, are predictable at the start of the influenza season. Vaccination cost includes the cost of the vaccine, its administration, and worker time spent being vaccinated. We found vaccination attractive only when administered in a lowcost site, such as an employee health service. Table 4 Annual effect (costs) of four influenza vaccination and treatment strategies on a cohort of 1000 healthy adult workers, averaged over 10 years Strategy Total Cost Lost Productivity Physician Office Visits Cost ($) Antiviral Drugs Rapid Testing Vaccine* Complications† Amantadine therapy No intervention Rapid test-oseltamivir Annual vaccination 234,000 236,000 237,000 239,000 219,000 222,000 219,000 216,000 12,500 12,500 12,500 11,400 250 0 1230 0 0 0 2280 0 0 0 0 9870 1840 1840 1820 920 *Vaccine and administration, not including worker time for administration. †Complications of influenza include sinusitis, pneumonia, and hospitalization. 74 The American Journal of Medicine, Vol 118, No 1, January 2005 Table 5 Sensitivity analysis* Variable Baseline Value (Range) Probability of physician visit within 48 hours Cost of a workday ($) Workdays lost per episode of influenza Workdays saved by a course of antiviral therapy Utility of a day of influenza Cost of vaccination ($) 0.4 177 1.9 1.0 0.6 21.06 (0.1–0.9) (0–500) (0.5–4.0) (0.8–1.2) (0–1) (16–26) Threshold Value† Range of Marginal Cost-effectiveness‡ ($/Quality-Adjusted Day Gained) — 455 2.44 — — 16.42 74–227 Cost-saving - 186 Cost-saving - 6300 88–145 47–1780 Cost-saving - 236 *Only variables that changed the marginal cost-effectiveness by at least 30% are presented. †Threshold at which annual vaccination becomes cost saving as compared with all other strategies. ‡Marginal cost-effectiveness as compared with amantadine therapy. The number of workdays missed as a result of influenza can be predicted based on worker demographic characteristics. Male workers and those in high-paying or demanding jobs report missing fewer workdays due to influenza.25,48 In 1996, female workers under the age of 44 years reported three times as many workdays lost due to influenza as did male employees aged 45 years or older. This partially explains why one randomized trial found vaccination to be costly, even when the vaccine was highly effective.6 In that study, workers were predominantly male, and three quarters had annual incomes of more than $70,000. Participants missed an average of 0.79 workdays per influenza-like illness. Similar results have been reported in studies of healthcare workers.7,49,50 In contrast, studies of factory and service workers found that these employees missed between 2.8 and 4.9 workdays per illness.51,52 Because worker age, sex, and compensation are known before the influenza season, these factors could be used in choosing workers for vaccination. Unlike worksite-specific determinants, influenza season– specific determinants, such as the incidence of influenza and the degree of match between the vaccine and circulating viruses, vary from year to year and are unknown at the beginning of the influenza season. For this reason, we relied on CDC reports of national influenza activity and circulating viral strains to predict how vaccination programs would have fared from 1992 to 2002. In years when influenza was prevalent and the vaccine well-matched, vaccination was inexpensive or even cost saving. When the match was poor or influenza was uncommon, vaccination was very expensive. On average, the cost-effectiveness of vaccination compares favorably with other commonly accepted interventions, provided the cost of vaccination is low.53 Given the variability in both worksite- and influenza season–specific determinants, it is not surprising that previous cost-effectiveness analyses have yielded discordant results. Although all investigators have found that vaccination improves health, the economic effect is less certain. Large, randomized placebo-controlled trials have only added to the controversy. One trial found that vaccination saved $47 per person vaccinated,17 whereas another, conducted over two consecutive influenza seasons, found that vaccination increased societal costs by $11 to $66 per person vaccinated, 100 Value of a Workday ($) 400 Annual Vaccination 300 Amantadine Therapy 200 X 100 0 1.0 +$5 -$5 No Intervention 1.5 2.0 2.5 3.0 3.5 4.0 Workdays Lost Per Episode of Influenza Illness Figure 3 Least costly influenza strategy as a function of worksite-specific variables. The graph is divided into zones, representing the least costly strategy. The solid line represents the thresholds among strategies in the base case; the dashed lines show how the threshold changes if the cost of vaccination is increased (large dashes) or decreased (small dashes) by $5. The X represents the base case values. Proportion of Monte Carlo Iterations Where Strategy Is Favored (%) 500 90 80 70 60 Annual Vaccination Test-Oseltamivir Amantadine 50 40 30 20 10 0 0 200 400 600 800 1000 Willingness to Pay ($/Quality-Adjusted Day Gained) Figure 4 Monte Carlo probabilistic sensitivity analysis for healthy workers over a period of 10 years. Each line shows the proportion of simulations for which that strategy would be optimal for each willingness-to-pay value. Rothberg and Rose Vaccination versus Treatment of Influenza depending on the year.6 Neither study considered the possibility of treatment with antiviral drugs. The use of antiviral drugs for influenza has been shown to be economically reasonable in working adults.14,15,54 Obviously, the greater the probability that the patient with influenza-like illness actually has influenza, the more cost-effective the use of antiviral medication. Based on the WHO surveillance criteria, even at the peak of the influenza season, the proportion of isolates testing positive for influenza never exceeds 33%.14 However, when influenza is locally prevalent, the WHO criteria of fever and cough have a positive predictive value of greater than 70%.55,56 The cost-effectiveness of antiviral therapy is also dependent on patients seeking medical attention within 48 hours of symptom onset. Although we assumed that only 40% of employees would seek attention, this variable is potentially within the employer’s control. Encouraging employees to seek prompt medical attention for fever and cough during the influenza season appears to save money, even for patients who have already been vaccinated, especially in years when the vaccine is not well matched to circulating strains of influenza. If patients seek medical attention after 48 hours, the cost-effectiveness of empiric amantadine will resemble no intervention, because no drugs will be dispensed. Whether the benefit of vaccination justifies the cost depends on the perspective of the payer. Although cost-effectiveness analyses are typically conducted from the societal perspective, society rarely pays for the interventions. For the employer whose goal is to minimize costs, vaccination makes sense only if worksite-specific variables favor vaccination. Health insurers or public payers who strive to provide cost-effective care should consider vaccinating healthy adults if vaccination costs less than $10, as in our base case. Live attenuated influenza vaccine is currently too expensive to consider for healthy adult workers. If it could be administered for under $10, it would offer a reasonable alternative to the injectable vaccine. Our analysis has limitations. By not including a pandemic year, we almost certainly underestimated the benefits of vaccination. In such years, attack rates may be much higher and mortality in healthy adults rises sharply,11 making vaccination cost saving. Conversely, we also assumed that vaccination side effects are mild. While in most years this is true, in 1976 the rate of postvaccination GuillainBarré syndrome was 10 times higher than usual. We also failed to include the effects of vaccination on nonvaccinated workers. Decreasing influenza among vaccinated workers may provide some protection to nonvaccinated colleagues and family members as well. In health care facilities, vaccinating workers protects patients.57 Moreover, not all workers are young and healthy. Instituting workplace vaccination could benefit workers with chronic illness who might not otherwise comply with current recommendations for vaccination. 75 After 20 years of data collection and public health policy analysis, experts agree that vaccination benefits healthy adults, but there is still no consensus on whether the benefit justifies the cost. Because all strategies produce similar outcomes, small variations in influenza rate, vaccine efficacy, or worker characteristics may tip the balance for or against vaccination. Although these factors are quantifiable, they are neither constant from year to year nor from place to place. Thus, additional randomized clinical trials cannot answer the broader question. Vaccination cannot be considered cost-effective under all circumstances, nor can it be generally rejected. Individual persons, health plans, and employers must decide on the basis of their specific circumstances, namely the cost of vaccination and the quantity of work lost due to influenza illness. For many healthy workers, annual vaccination is reasonable economically, but it will not save money. At a minimum, prudent use of amantadine, as compared with no intervention, should consistently reduce both days of illness and costs. Appendix Estimating the weekly incidence of influenza and influenza-like illness is challenging because both vary from week to week. During the influenza season, the Centers for Disease Control and Prevention (CDC) reports the number of influenza-like illness specimens received from its sentinel sites each week, as well as the proportion that test positive for influenza. Assuming that the number of specimens collected is proportional to the number of cases of influenzalike illness, we were able to calculate the number of influenza cases for any given week y by the formula iy ⫽ b * (wy/a) * py, where iy ⫽ number of influenza cases for week y, b ⫽ baseline rate of influenza-like illness, wy ⫽ the number of influenza-like illness cases in week y, a ⫽ the average number of influenza-like illness cases that year, and py ⫽ the percentage of influenza-like illness cases caused by influenza in week y. All values are reported by the CDC except for b. Empirically varying b results in different predicted annual rates of influenza illness. When b ⫽ 2% per week, the average predicted annual influenza attack rate is 8.6%, with annual rates ranging from 6% to 10%, in keeping with estimates by others.1,6,13 References 1. Szucs T: The socio-economic burden of influenza. J Antimicrob Chemother 1999;44(suppl B):11–15 2. Nichol KL, Baken L, Wuorenma J, Nelson A: The health and economic benefits associated with pneumococcal vaccination of elderly persons with chronic lung disease. Arch Intern Med 1999;159:2437– 2442 3. Nichol KL, Margolis KL, Wuorenma J, Von Sternberg T: The efficacy and cost effectiveness of vaccination against influenza among elderly persons living in the community. N Engl J Med 1994;331:778 –784 76 4. Davis JW, Lee E, Taira DA, Chung RS: Influenza vaccination, hospitalizations, and costs among members of a Medicare managed care plan. Med Care 2001;39:1273–1280 5. Nordin J, Mullooly J, Poblete S, et al: Influenza vaccine effectiveness in preventing hospitalizations and deaths in persons 65 years or older in Minnesota, New York, and Oregon: data from 3 health plans. J Infect Dis 2001;184:665– 670 6. Bridges CB, Thompson WW, Meltzer MI, et al: Effectiveness and cost-benefit of influenza vaccination of healthy working adults: a randomized controlled trial. JAMA 2000;284:1655–1663 7. Weingarten S, Staniloff H, Ault M, Miles P, Bamberger M, Meyer RD: Do hospital employees benefit from the influenza vaccine? A placebocontrolled clinical trial. J Gen Intern Med 1988;3:32–37 8. Campbell DS, Rumley MH: Cost-effectiveness of the influenza vaccine in a healthy, working-age population. J Occup Environ Med 1997;39:408 – 414 9. Cost-effectiveness of flu vaccine. Congressional Office of Technology Assessment. Conn Med 1982;46:607 10. Muennig PA, Khan K: Cost-effectiveness of vaccination versus treatment of influenza in healthy adolescents and adults. Clin Infect Dis 2001;33:1879 –1885 11. Simonsen L, Clarke MJ, Schonberger LB, Arden NH, Cox NJ, Fukuda K: Pandemic versus epidemic influenza mortality: a pattern of changing age distribution. J Infect Dis 1998;178:53– 60 12. Demicheli V, Rivetti D, Deeks J, Jefferson T: Vaccines for preventing influenza in healthy adults. Cochrane Database Syst Rev Volume 2; 2002 13. Nichol KL: Cost-benefit analysis of a strategy to vaccinate healthy working adults against influenza. Arch Intern Med 2001;161:749 –759 14. Centers for Disease Control and Prevention. Reports and Surveillance Methods In The United States: Current U.S. Flu Report. 2002. Available at: http://www.cdc.gov/ncidod/diseases/flu/ weeklychoice.htm. Accessed May 28, 2002 15. Rothberg MB, He S, Rose DN: Management of influenza symptoms in healthy adults. J Gen Intern Med 2003;18:808 – 815 16. Smith KJ, Roberts MS: Cost-effectiveness of newer treatment strategies for influenza. Am J Med 2002;113:300 –307 17. Nichol KL, Lind A, Margolis KL, et al: The effectiveness of vaccination against influenza in healthy, working adults. N Engl J Med 1995;333:889 – 893 18. Treanor JJ, Hayden FG, Vrooman PS, et al: Efficacy and safety of the oral neuraminidase inhibitor oseltamivir in treating acute influenza: a randomized controlled trial. US Oral Neuraminidase Study Group. JAMA 2000;283:1016 –1024 19. Kaiser L, Henry D, Flack NP, Keene O, Hayden FG: Short-term treatment with zanamivir to prevent influenza: results of a placebocontrolled study. Clin Infect Dis 2000;30:587–589 20. Nicholson KG, Aoki FY, Osterhaus AD, et al: Efficacy and safety of oseltamivir in treatment of acute influenza: a randomised controlled trial. Neuraminidase Inhibitor Flu Treatment Investigator Group. Lancet 2000;355:1845–1850 21. Neuzil KM, Reed GW, Mitchel EF Jr, Griffin MR: Influenza-associated morbidity and mortality in young and middle-aged women. JAMA 1999;281:901–907 22. Barker WH, Mullooly JP: Impact of epidemic type A influenza in a defined adult population. Am J Epidemiol 1980;112:798 – 811 23. Simonsen L, Fukuda K, Schonberger LB, Cox NJ: The impact of influenza epidemics on hospitalizations. J Infect Dis 2000;181:831– 837 24. Nichol KL, Mendelman PM, Mallon KP, et al: Effectiveness of live, attenuated intranasal influenza virus vaccine in healthy, working adults: a randomized controlled trial. JAMA 1999;282:137–144 25. Olsen GW, Burris JM, Burlew MM, et al: Absenteeism among employees who participated in a workplace influenza immunization program. J Occup Environ Med 1998;40:311–316 26. Cox FM, Cobb MM, Chua WQ, McLaughlin TP, Okamoto LJ: Cost of treating influenza in emergency department and hospital settings. Am J Manag Care 2000;6:205–214 The American Journal of Medicine, Vol 118, No 1, January 2005 27. Nichol KL, Margolis KL, Lind A, et al: Side effects associated with influenza vaccination in healthy working adults. A randomized, placebo-controlled trial. Arch Intern Med 1996;156:1546 –1550 28. Lasky T, Terracciano GJ, Magder L, et al: The Guillain-Barre syndrome and the 1992-1993 and 1993-1994 influenza vaccines. N Engl J Med 1998;339:1797–1802 29. Jefferson T, Demicheli V, Rivetti D, Deeks J: Cochrane reviews and systematic reviews of economic evaluations. Amantadine and rimantadine in the prevention and treatment of influenza. Pharmacoeconomics 1999;16(suppl 1):85– 89 30. Wingfield WL, Pollack D, Grunert RR: Therapeutic efficacy of amantadine HCl and rimantadine HCl in naturally occurring influenza A2 respiratory illness in man. N Engl J Med 1969;281:579 – 584 31. Van Voris LP, Betts RF, Hayden FG, Christmas WA, Douglas RG Jr: Successful treatment of naturally occurring influenza A/USSR/77 H1N1. JAMA 1981;245:1128 –1131 32. Dolin R, Reichman RC, Madore HP, Maynard R, Linton PN, WebberJones J: A controlled trial of amantadine and rimantadine in the prophylaxis of influenza A infection. N Engl J Med 1982;307:580 –584 33. Hayden FG, Atmar RL, Schilling M, et al: Use of the selective oral neuraminidase inhibitor oseltamivir to prevent influenza. N Engl J Med 1999;341:1336 –1343 34. Hayden FG, Treanor JJ, Fritz RS, et al: Use of the oral neuraminidase inhibitor oseltamivir in experimental human influenza: randomized controlled trials for prevention and treatment. JAMA 1999;282:1240 – 1246 35. 2002 Mosby’s Drug Consult. St. Louis, Missouri: Mosby, Inc.; 2002 36. U.S. Bureau of Labor Statistics: 2000 National Occupational Employment and Wage Estimates Healthcare Practitioners and Technical Occupations. Available at: http://www.bls.gov/oes/2000/ oes_29He.htm. Accessed October 2, 2002. 37. 2001 Drug Topics Red Book. Montvale, New Jersey: Medical Economics, Inc; 2001 38. Crane M: What you charge vs what you get. Med Econ 2001;78:50 – 52,55-57 39. The MEDSTAT Group: 2000 DRG Guide: Descriptions and Normative Values. Ann Arbor, Michigan: MEDSTAT Group; 2001 40. Meltzer MI, Cox NJ, Fukuda K: The economic impact of pandemic influenza in the United States: priorities for intervention. Emerg Infect Dis 1999;5:659 – 671 41. U.S. Bureau of Labor Statistics: Employer Costs for Employee Compensation—March 2001. Available at: ftp://ftp.bls.gov/pub/special. requests/ocwc/ect/ececrlse.pdf. Accessed May 28, 2002. 42. Kaiser L, Keene ON, Hammond JM, Elliott M, Hayden FG: Impact of zanamivir on antibiotic use for respiratory events following acute influenza in adolescents and adults. Arch Intern Med 2000;160:3234 – 3240 43. Brammer L, Fukuda K, Arden N, et al: Influenza surveillance—United States, 1992-93 and 1993-94. Mor Mortal Wkly Rep CDC Surveill Summ. 1997;46:1–12 44. Brammer TL, Izurieta HS, Fukuda K, et al: Surveillance for influenza—United States, 1994-95, 1995-96, and 1996-97 seasons. Mor Mortal Wkly Rep CDC Surveill Summ 2000;49:13–28 45. Griffin AD, Perry AS, Fleming DM: Cost-effectiveness analysis of inhaled zanamivir in the treatment of influenza A and B in high-risk patients. Pharmacoeconomics 2001;19:293–301 46. Gold M, Siegel J, Russell L, Weinstein M, eds: Cost-Effectiveness in Health and Medicine. New York, New York: Oxford University Press; 1996 47. U.S. Bureau of Labor Statistics. Consumer Price Index—All Urban Consumers. Available at: http://data.bls.gov/cgi-bin/surveymost?cu. Accessed May 28, 2002 48. Adams P, Hendershot G, Marano M: Current estimates from the National Health Interview Survey, 1996. Vital Health Stat 10. 1999; 200:55 Rothberg and Rose Vaccination versus Treatment of Influenza 49. Saxen H, Virtanen M: Randomized, placebo-controlled double blind study on the efficacy of influenza immunization on absenteeism of health care workers. Pediatr Infect Dis J 1999;18:779 –783 50. Wilde JA, McMillan JA, Serwint J, Butta J, O’Riordan MA, Steinhoff MC: Effectiveness of influenza vaccine in health care professionals: a randomized trial. JAMA 1999;281:908 –913 51. Keech M, Scott AJ, Ryan PJ: The impact of influenza and influenzalike illness on productivity and healthcare resource utilization in a working population. Occup Med (Lond) 1998;48:85–90 52. Kumpulainen V, Makela M: Influenza vaccination among healthy employees: a cost-benefit analysis. Scand J Infect Dis 1997;29:181–185 53. Mark DB, Hlatky MA, Califf RM, et al: Cost effectiveness of thrombolytic therapy with tissue plasminogen activator as compared with streptokinase for acute myocardial. N Engl J Med 1995;332:1418 –1424 77 54. Lee PY, Matchar DB, Clements DA, Huber J, Hamilton JD, Peterson ED: Economic analysis of influenza vaccination and antiviral treatment for healthy working adults. Ann Intern Med 2002;137: 225–231 55. Boivin G, Hardy I, Tellier G, Maziade J: Predicting influenza infections during epidemics with use of a clinical case definition. Clin Infect Dis 2000;31:1166 –1169 56. Zambon M, Hays J, Webster A, Newman R, Keene O: Diagnosis of influenza in the community: relationship of clinical diagnosis to confirmed virological, serologic, or molecular detection of influenza. Arch Intern Med 2001;161:2116 –2122 57. Potter J, Stott DJ, Roberts MA, et al: Influenza vaccination of health care workers in long-term-care hospitals reduces the mortality of elderly patients. J Infect Dis 1997;175:1– 6
© Copyright 2026