Needle-Free-Injections-Medical-Devices-Pipeline-Assessment-2016
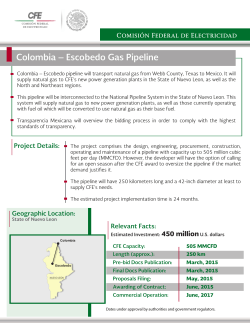
Needle Free Injections - Medical Devices Pipeline Assessment, 2016 No of Pages – 64 Publishing Date - May 1, 2016 Needle Free Injections - Medical Devices Pipeline Assessment, 2016 Summary GlobalData's Medical Devices sector report, “Needle Free Injections - Medical Devices Pipeline Assessment, 2016" provides an overview of Needle Free Injections currently in pipeline stage. The report provides comprehensive information on the pipeline products with comparative analysis of the products at various stages of development. The report reviews major players involved in the pipeline product development. It also provides information about clinical trials in progress, which includes trial phase, trial status, trial start and end dates, and, the number of trials for the key Needle Free Injections pipeline products. This report is prepared using data sourced from in-house databases, secondary and primary research by GlobalData's team of industry experts. *Note: Certain sections in the report may be removed or altered based on the availability and relevance of data in relation to the equipment type. Scope - Extensive coverage of the Needle Free Injections under development - The report reviews details of major pipeline products which includes, product description, licensingand collaboration details and other developmental activities - The report reviews the major players involved in the development of Needle Free Injections and listall their pipeline projects - The coverage of pipeline products based on various stages of development ranging from Early Development to Approved / Issued stage - The report provides key clinical trial data of ongoing trials specific to pipeline products - Recent developments in the segment / industry Reasons to buy The report enables you to - Formulate significant competitor information, analysis, and insights to improve R&D strategiesIdentify emerging players with potentially strong product portfolio and create effective counterstrategies to gain competitive advantage - Identify and understand important and diverse types of Needle Free Injections under development - Develop market-entry and market expansion strategies - Plan mergers and acquisitions effectively by identifying major players with the most promisingpipeline - In-depth analysis of the product’s current stage of development, territory and estimated launch date Ask for Discount at – http://www.marketreportsworld.com/enquiry/request-discount/10160235 1 Table of Contents 1 Table of Contents 2 1.1 List of Tables 4 1.2 List of Figures 5 2 Introduction 6 2.1 Needle Free Injections Overview 6 3 Products under Development 7 3.1 Needle Free Injections - Pipeline Products by Stage of Development 7 3.2 Needle Free Injections - Pipeline Products by Territory 8 3.3 Needle Free Injections - Pipeline Products by Regulatory Path 9 3.4 Needle Free Injections - Pipeline Products by Estimated Approval Date 10 4 Needle Free Injections - Pipeline Products under Development by Companies 11 4.1 Needle Free Injections Companies - Pipeline Products by Stage of Development 11 4.2 Needle Free Injections - Pipeline Products by Stage of Development 12 5 Needle Free Injections Companies and Product Overview 13 5.1 B. Braun Melsungen AG Company Overview 13 5.1.1 B. Braun Melsungen AG Pipeline Products & Ongoing Clinical Trials Overview 13 5.2 Bioject Medical Technologies Inc. Company Overview 15 5.2.1 Bioject Medical Technologies Inc. Pipeline Products & Ongoing Clinical Trials Overview 15 5.3 Boston Scientific Corporation Company Overview 21 5.3.1 Boston Scientific Corporation Pipeline Products & Ongoing Clinical Trials Overview 21 5.4 Crossject SA Company Overview 23 5.4.1 Crossject SA Pipeline Products & Ongoing Clinical Trials Overview 23 5.5 Enable Injections, LLC Company Overview 29 5.5.1 Enable Injections, LLC Pipeline Products & Ongoing Clinical Trials Overview 29 5.6 Eternity Healthcare Inc. Company Overview 31 5.6.1 Eternity Healthcare Inc. Pipeline Products & Ongoing Clinical Trials Overview 31 5.7 EuroJet Medical Kft. Company Overview 32 5.7.1 EuroJet Medical Kft. Pipeline Products & Ongoing Clinical Trials Overview 32 5.8 Glide Pharmaceutical Technologies Limited Company Overview 33 5.8.1 Glide Pharmaceutical Technologies Limited Pipeline Products & Ongoing Clinical Trials Overview 33 5.9 Medical International Technologies (Mit Canada) Inc. Company Overview 35 5.9.1 Medical International Technologies (Mit Canada) Inc. Pipeline Products & Ongoing ClinicalTrials Overview 35 5.10 PharmaJet, Inc. Company Overview 41 5.10.1 PharmaJet, Inc. Pipeline Products & Ongoing Clinical Trials Overview 41 5.11 Teva Pharmaceutical Industries Limited Company Overview 42 5.11.1 Teva Pharmaceutical Industries Limited Pipeline Products & Ongoing Clinical Trials Overview 42 5.12 Vaxxas Pty. Ltd. Company Overview 44 5.12.1 Vaxxas Pty. Ltd. Pipeline Products & Ongoing Clinical Trials Overview 44 6 Needle Free Injections- Recent Developments 45 6.1 Apr 19, 2016: Rodney D. Altman, MD Joins Valeritas Board of Directors 45 6.2 Mar 21, 2016: ICU Medical Enters into Distribution Agreement with B. Braun Melsungen 45 6.3 Mar 15, 2016: Aradigm Announces Fourth Quarter 2015 and Full Year Financial Results 45 6.4 Mar 14, 2016: Inovio Pharma to Acquire Assets of Bioject Medical Technologies for USD5.5 Million 47 6.5 Mar 10, 2016: Zogenix Provides Corporate Update and Reports Fourth Quarter and Full-Year 2015 Financial Results 47 6.6 Jan 07, 2016: Corporate Flu Clinics Offering PharmaJet Needle-Free Flu Shots 48 6.7 Dec 09, 2015: Crossject Enters into Agreement with Xi’an Xintong Pharma 49 6.8 Nov 16, 2015: Allmed Acquires Majority Stake in Pronefro-Produtos 49 6.9 Nov 12, 2015: Intermountain Healthcare Enters into Agreement with Velano Vascular 49 6.10 Nov 09, 2015: Zogenix Provides Update and Reports Third Quarter 2015 Financial Results 49 6.11 Oct 06, 2015: Rx Safes Enters into Distribution Agreement with Mika Medical and MK Global 51 6.12 Oct 05, 2015: Medicom receives the growth award - Borsen Gazelle 2015 52 6.13 Sep 14, 2015: Crossject Enters into Agreement with Sayre Therapeutics 52 6.14 Sep 09, 2015: Enable Injections Completes Its First-In-Human Wearable Injector Pilot Trial 52 6.15 Aug 20, 2015: PharmaJet Needle-Free Injector Becomes Part of the American Pharmacists Association Educational Curriculum 53 6.16 Aug 13, 2015: Aradigm Announces Second Quarter 2015 Financial Results 53 6.17 Aug 10, 2015: Zogenix Provides Corporate Update and Reports Second Quarter 2015 Financial Results 54 6.18 Jul 28, 2015: Zogenix Raises USD98.3 Million in Public Offering of Shares 56 6.19 Jul 16, 2015: CROSSJECT confirms the development plan for ZENEO Sumatriptan 56 6.20 Jul 06, 2015: Zogenix Expands Senior Leadership Team to Further Drive Strategic Focus on CNS Disorders and Orphan Drug Development 57 6.21 Jun 22, 2015: Biocorp Production Raises USD9.8 Million in IPO 57 6.22 Jun 22, 2015: Owen Mumford Expands Global Foot Print to Support Asia Pacific 58 6.23 May 11, 2015: Aradigm Announces First Quarter 2015 Financial Results 58 6.24 May 11, 2015: Zogenix Provides Corporate Update and Reports First Quarter 2015 Financial Results 59 7 Appendix 61 7.1 Methodology 61 7.2 About GlobalData 63 7.3 Contact Us 64 7.4 Disclaimer 64 Report Details – Date of Publishing - May 1, 2016 No of Pages – 64 Single User PDF License – $2500 Corporate License – $7500 Buy Now - http://www.marketreportsworld.com/purchase/10160235 About Market Reports World: Market Reports World is the credible source for gaining the market research reports that will exponentially accelerate your business. We are among the leading report resellers in the business world committed towards optimizing your business. The reports we provide are based on a research that covers a magnitude of factors such as technological evolution, economic shifts and a detailed study of market segments. Contact– Mr. Ameya Pingaley Market Reports World +1-408 520 9750 Email – [email protected] www.marketreportsworld.com Powered by TCPDF (www.tcpdf.orPowered by TCPDF (www.tcpdf.orgg) )
© Copyright 2026