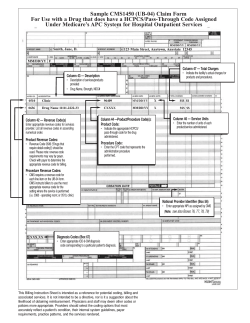
CMS Tells States To Reimburse Plans For Health Insurance Provider Fee
InsideHealthPolicy.com’s Inside CMS exclusive news on the most powerful agency in health care Vol. 17, No. 42 - October 16, 2014 CMS Tells States To Reimburse Plans For Health Insurance Provider Fee CMS says that states should reimburse Medicaid managed care plans for the Affordable Care Act’s health insurance provider fee, and says that the fee itself should be incorporated into plans’ capitation rates. However, as the firm JP Morgan notes, the agency leaves some “wiggle room” on whether states should also factor in other potential effects of the fee, such as its non-deductibility status. Medicaid Health Plans of America President Jeff Myers said CMS’ recently released frequently asked questions document provides certainty for plans, particularly in states that hadn’t yet agreed to cover those fees. Myers said the plans are gratified CMS decided to move forward with the FAQ, and the release of the FAQ and the 2015 Managed Care Rate Setting Consultation guide suggest that CMS would like to play a more involved role in rate setting for managed care. Since plans have been asking for more transparency around rate setting, CMS’ involvement could be a net positive, Myers said. The Government Accountability Office has also said that CMS’ oversight of the rate-setting process needs improvement. A JP Morgan investors note points out that CMS’ FAQ, released Tuesday (Oct. 7), explicitly states for the first time that the agency believes states should reimburse MCOs for the continued on page 10 New Hepatitis C Drug’s Cost Lower Than Expected, Fuels Rx-Pricing Debate Gilead Science’s revelation Friday (Oct. 10) that it will charge $94,500 for a 12-week course of its newly approved hepatitis C drug Harvoni, though less than some market analysts had expected, stoked the national drug pricing debate, with drug price-control advocate promptly urging Congress to stem a brewing crises of what they called “excessive pricing” by drug makers. Medicaid health plans simultaneously released a report detailing prescribing and coverage restrictions states have employed to manage the high cost of Gilead’s first hepatitis C drug, Sovaldi, and warning that states will increasingly use such methods as more expensive drugs hit the market. National Coalition on Health Care President John Rother wrote an open continued on page 12 AMA Sends CMS ‘Blueprint’ For Revamping EHR Meaningful Use Program The American Medical Association is pressing CMS to significantly relax rules for the meaningful use of electronic health records going forward, including abandoning what AMA calls the current program’s “all-or-nothing” approach under which providers will either receive an incentive or face a penalty under Stage 2 of the program as the agency prepares regulations for Stage 3. The group wrote to CMS Administrator Marilyn Tavenner and National Coordinator for Health Information Technology Karen DeSalvo Tuesday (Oct. 14) that the meaningful use program is at a critical crossroads where it is important to pause and assess what is working and what needs to be fixed to ensure the program’s success. continued on page 14 Legal fight over 340B orphan drug rule continues PhRMA Files New Lawsuit Challenging HRSA’s 340B Orphan Drug Rule The Pharmaceutical Research and Manufacturers of America filed a new lawsuit Thursday (Oct. 9) challenging the Health Resources and Services Administration’s interpretive rule on the 340B orphan drug exclusion, as the group was advised to do by a federal judge in late August if it wanted to contest the agency’s policy. At issue is HRSA’s stance that the ACA’s exemption of orphan drugs from the 340B program’s drug discounts for safety net hospitals doesn’t apply when the drugs are used to treat conditions not part of their orphan indication. In his Aug. 27 ruling federal District Court Judge Rudolph Contreras said continued on page 16 MedPAC Weighs Proposals To Shift Rx Plans’ Reinsurance, Risk Corridors Several congressional Medicare advisers said Thursday (Oct. 9) that the government provides drug plans more protection than they need but added that they aren’t sure how to change reinsurance and risk corridor policies in part because it is unclear how policy shifts would affect so-called low-income subsidy beneficiaries. The policy options that the Medicare Payment Advisory Commission began discussing this week will likely be included in a report to Congress later this year, but it’s not clear whether MedPAC will make formal recommendations that soon. MedPAC researchers said Congress gave redundant protections to plans when they created the Part D drug benefit because plans worried that only sicker beneficiaries would enroll. However, the market for prescription drug plans is now well established — most beneficiaries have about 30 stand-alone plans from which to choose, and Medicare Advantage plans often provide drug coverage, too — so it’s probably time to make plans accept more risk, several commissioners said. Drug plans receive three types of protections. Medicare risk adjusts subsidies for beneficiaries to account for the higher spending on sicker patients, and Medicare pays bigger subsidies for low-income patients who pay little to nothing out-of-pocket. Medicare also pays 80 percent of the cost of every beneficiaries’ drug spending once they enter catastrophic coverage. Plans pay 15 percent and beneficiaries pay 5 percent. Both of those protections are for individual enrollees. Then, Medicare insures plans against aggregate drug spending with risk corridors. When plans underestimate total drug spending in their bids, the risk corridor policy pays them back a portion of the amount over their estimates. Plans are responsible for the first 5 percent over their estimates. Medicare pays for half of spending between 105 percent and 110 percent of spending, and above that Medicare pays 80 percent back to plans. Likewise, when annual plan bids are based on overestimates of spending, plans pay Medicare back. Plans consistently overestimate spending, set bids too high and end up paying Medicare back a portion of the subsidies they receive. Commissioners note that seniors are the only entity to truly lose out in this scenario because they pay higher premiums and are paid back nothing. Commissioners said they don’t know why plans consistently bid too high or why CMS accepts those bids. MedPAC researchers plan to talk with plans about why in the past several years they have nearly always overbid. Risk corridors accounted for 19 percent of Medicare’s drug spending in 2007 and last year they accounted for 31 percent of spending. MedPAC researcher Rachel Schmidt, who helped put together the original spending projections on risk corridors while at the Congressional Budget Office about a decade ago, said CBO did not anticipate that the risk corridors would account for such a large portion of Medicare spending on drugs. However, commissioners struggled to predict how changes to reinsurance and risk corridors would affect beneficiaries. At one point, several MedPAC members talked about whether widening risk corridors would likely lead to higher or Hot Documents Now Available on InsideHealthPolicy.com The following new documents are available on InsideHealthPolicy.com, our new online health news service. Subscribers to InsideHealthPolicy.com also have access to hundreds of other health-related documents, daily news updates, and a searchable archive of back issues. HHS: 2015 Medicare Part B Premiums, Deductibles To Remain Same As Last Two Years PhRMA Sues HRSA Again Over 340B Orphan Drug Policy HHS Announces $238 Million Investment In Primary Care Workforce CMTP Releases White Paper On Enhanced Recovery For Surgical Patients CMS Publishes 2015 Star Ratings For MA, Drug Plans New Administration FAQ Discusses “Reference Pricing” Standards For Non-Grandfathered Group Plans FDA Approves First Combination Pill To Treat Hepatitis C Campaign For Sustainable Rx Pricing Blasts Gilead’s Pricing Of Harvoni Medicaid Health Plans Study Details How States Are Addressing Sovaldi’s Costs AARP Urges Congress To Extend CHIP Funding Through 2019 Plaintiff’s Reply To Government’s Opposition Of Supreme Court Review In Case Regarding FFM Subsidies CMS Releases Education Materials On QHP Renewal Process AMA Asks CMS For More Flexibility In Meaningful Use Program CMS Announces New ACO Investment Model For Rural, Underserved Areas Not an online subscriber? Inside CMS subscribers can get a free, two-week trial to InsideHealthPolicy.com by calling 1-800-424-9068 or signing up at http://www.insidehealthpolicy.com/health_signup.asp. 2 INSIDE CMS — www.InsideHealthPolicy.com — October 16, 2014 lower premiums, and they didn’t come to a conclusion. MedPAC is especially worried about beneficiaries for whom plans get low-income subsidies. Medicare pays low-income subsidies for about one-third of Part D enrollees, and these are the most vulnerable beneficiaries. About 75 percent of low-income subsidy beneficiaries are in stand-alone prescription drug plans and the other 25 percent are enrolled in Medicare Advantage prescription drug plans. Changes to risk sharing could affect incentives to enroll these beneficiaries, commissioners said. Several commissioners said they believe the corridors should be widened so plans are on the hook for more when they underbid, but that also would mean they’d pay Medicare back less when they overbid. Commissioner Jack Hoadley, of the Georgetown University Health Policy Institute, said he leans toward widening risk corridors but he added that his thinking on the subject has changed somewhat in light of expectations that specialty drugs will drive up drug spending in the coming years. Commissioner also entertained lowering the reinsurance percentage that Medicare pays during catastrophic coverage, but said they didn’t know what mix of reinsurance and risk corridor policies to recommend. Commissioner Katherine Baicker, of Harvard School of Public Health, said plans should be able to buy private reinsurance. MedPAC Commissioner Glenn Hackbarth added that Medicare Advantage plans that offer drug coverage do not receive the same level of protection, yet they’ve not had a difficult time. Nevertheless, Hackbarth said the market is dynamic and he fears unintended consequences and wants to understand better the potential impacts of policy changes before the commission makes recommendations.—John Wilkerson CMS Steps Up Bid To Smooth Part D, Hospice Communication CMS is collecting feedback on a standardized prior authorization form for beneficiaries enrolled in hospice to help Part D (drug) plans and hospice providers communicate about beneficiaries’ medications, hospice enrollment and discharge. The agency says a uniform process will make prior authorization for hospice beneficiaries’ applicable Part D prescriptions more efficient, and one hospice lobbyist said a standardized form could help streamline the processes and simplify prior authorization. “We believe that the use of a uniform process and form will improve communication between hospice providers and Part D plans thereby enhancing the efficiency of the PA process, and will be to the overall benefit of Medicare beneficiaries,” the agency says in a memo. The standardized form is a tweaked version of form that the industry has been working on for over a year through the National Council of Prescription Drug Plans, according to Judi Lund Person with the National Hospice and Palliative Care Organization. CMS says the form is already in use by some Part D sponsors, and a supporting statement from the agency says CMS hopes to have the form available for use starting in January. Person said that one goal is to make the process for communication with Part D plans more uniform, and to streamline it. One beneficiary advocate, however, said problems could surface if prior authorization requests are not accepted once the form goes into effect because they are not in the standardized format. Person, however, said there’s no reason for a hospice not to use the standardized form. CMS had previously laid out a list of information that hospices needed to send to plans in order to complete prior authorization, and said that as long as the information was passed from a hospice to a Part D plan, the plan needed to accept it. The beneficiary advocate said the form essentially formalizes what was laid out in the agency’s previous memos. March CMS guidance laying out what information hospices needed to share with Part D plans also said plans should place all hospice beneficiaries’ drugs under prior authorization, as the hospices are responsible for paying for drugs related to all terminal and related conditions. The guidance also said it would be rare that Part D plans would be required to pay for a hospice beneficiaries’ drugs. After pushback from stakeholders, the agency in July instead limited Part D prior authorization requirements to four categories of prescription drugs — analgesics, anti-nauseants, laxatives, and anti-anxiety drugs. “The form would be used proactively (i.e., prior to the submission of a claim to Part D), to prevent a drug claim from rejecting at point-of-sale when a drug in any of the four classes is prescribed for a condition that is unrelated to a beneficiary’s terminal illness and/or related conditions and the beneficiary is prepared to procure the drug,” the CMS memo says. Another beneficiary advocate said this is overall a good step, and the hope is that beneficiary will not face any extra burden when trying to access necessary prescriptions due to the communication between hospices and Part D plans. However, there is come concern over CMS’ statement in the memo that if documentation is not submitted in advance, “the pharmacy will notify the beneficiary of the reject and may also notify the prescriber or the hospice.” The advocate says that the pharmacy should be required to notify the prescriber, the hospice or both so that the burden of doing so is not put on the beneficiary. In another reference, CMS appears to go a step further and says that the pharmacy should contact the beneficiary’s hospice provider if a drug is denied, and goes on to say that the pharmacy should contact the beneficiary’s prescriber only INSIDE CMS — www.InsideHealthPolicy.com — October 16, 2014 3 if the hospice provider is unknown and “other sources have been exhausted to identify the beneficiary’s hospice.” Person says that since CMS will now only apply prior authorization to the four classes of drugs, communicating a beneficiary’s change in hospice status will be just as important as communicating which drugs need prior authorization, if not more so. Chances are, prior authorization for those classes of drugs will not be used that often, Person says, but if a beneficiary is discharged from hospice, the form will be very important to keep that person from being turned away at the pharmacy counter. It will also be important to keep Part D plans and hospices from covering the same drug twice, she added. CMS also says that “[i]nforming the plan sponsor of the additional medications prescribed and designating the responsible financial party will assist the sponsor in better managing their beneficiary’s coverage and appropriate access to medication.” — Michelle M. Stein CMS Faces Glitches As It Ends Policies Linked To Citizen-Related Issues The federal marketplace is running into some back-end problems as it moves to terminate insurance policies for people with citizen-related inconsistencies, including generating more 834 transactions to insurers than needed and in some cases not terminating coverage when it should be, according to CMS slides on the terminations. An HHS official tells Inside Health Policy that the issues have not impacted any individuals. “We continue to work to fine tune the processes on how issuers receive information from the Marketplace regarding the status of their policyholders,” the spokesperson said. “As in this case, consumers are typically not impacted by these issues,” the official adds. According to the slides, in certain cases where multiple people in the family have an expiring inconsistency, “unexpected 834 transactions” were generated. The 834 transmissions contain enrollment data such as an individual’s social security number, their dependents and the plan that they picked, and in this case the transactions indicate that a plan was terminated, canceled or otherwise changed. The slides present an example in which the system generated four 834 transactions instead of two as was expected. The example represents an enrollee group of three people — John, the subscriber; Jane, the spouse; and a child — in which the two parents both have an inconsistency in their citizen-related data and thus their coverage will be terminated. In the example, the system first terminated the entire group, then created a new group with Jane as the subscriber plus the child. The system then canceled Jane’s coverage (it was canceled not terminated because the coverage had a future effective date) and created a new plan with the child as the subscriber. Termination refers to the practice of an insurer discontinuing an existing insurance policy, while cancellation refers to termination of a policy by an insurer prior to the policy’s effectuation. Additionally, CMS says that “proactive” quality assurance on the 834s turned up a unique scenario in which the consumer’s insurance may not be terminated when it should be. The document says this occurs when there are multiple consumers in an enrollment group and all have expiring inconsistencies. CMS offers no more details on this. The agency says it is currently validating findings from a “root cause analysis” and will apply a fix, and will “distribute additional 834 transactions if necessary.” CMS in August announced that it would be sending out terminating notices to about 310,000 people who had unresolved data matching issues — or situations in which the information on an application did not match existing records. The agency gave individuals a Sept. 5 deadline to send in requested documents or face plan terminations by Sept. 30. In addition to losing coverage, those individuals targeted could also be faced with repaying subsidies they received. The agency announced on Sept. 15 that the 310,000 number had been whittled down to 115,000 individuals. Despite advocates’ concerns about lack of language services and other issues that may have spurred confusion, the agency began the termination process as planned. On Sept. 30, the National Immigration Law Center and other immigration advocates filed an anti-discrimination complaint under Section 1557 of the ACA over HHS’ failure to correspond with consumers facing coverage terminations in languages other than English and Spanish. The groups also filed a Freedom of Information Act request in an effort to glean more information on the department’s decision-making process. —Amy Lotven SUBSCRIPTIONS: 703-416-8500 or 800-424-9068 [email protected] NEWS OFFICE: 703-416-8577 Fax: 703-416-8543 [email protected] 4 Health Group Publisher: Chief Editor: Managing Editor: Associate Editor: Contributing Editor: Donna Haseley ([email protected]) John Wilkerson ([email protected]) Michelle M. Stein ([email protected]) Todd Allen Wilson ([email protected]) Amy Lotven ([email protected]) Production Manager: Production Specialists: Lori Nicholson Daniel Arrieta, Michelle Moodhe Inside CMS is published every Thursday by Inside Washington Publishers, P.O. Box 7167, Ben Franklin Station, Washington, DC 20044. Subscription rates: $705 per year in U.S. and Canada; $755 per year elsewhere (air mail). © Inside Washington Publishers, 2014. All rights reserved. Contents of Inside CMS are protected by U.S. copyright laws. No part of this publication may be reproduced, transmitted, transcribed, stored in a retrieval system, or translated into any language in any form or by any means, electronic or mechanical, without written permission of Inside Washington Publishers. INSIDE CMS — www.InsideHealthPolicy.com — October 16, 2014 CMS Assesses Parallel Review; Device Industry Urges Continued Funding CHICAGO — A CMS official told device industry representatives the agency plans to evaluate whether changes or improvements are needed to the FDA/CMS parallel review program, as the two agencies laud their first parallel review product approval and enter the last year of the pilot in 2015. Industry supporters of the program worry that its funding could end, and Exact Sciences Corporation — the one company that saw success with the program — is trying to build support on Capitol Hill to continue the effort. Several device industry stakeholders praised the increased interaction and transparency with FDA and CMS generally but cited coverage as a market barrier that still looms, speaking at the Advanced Medical Technology Association’s medtech conference last week. Another company, Covidien, signaled it is trying to get one of its new technologies into the program, but a Covidien official cited government resource constraints and industry fear of engaging with the two agencies as hurdles to the initiative’s ultimate success. A CMS official said the agency has had a positive experience with the parallel review program but will look into whether it should be modified or improved. She told the device industry audience that CMS is still learning when it should become involved in the parallel review process. “If we’re too early, I’m not sure that’s the best use of everybody’s time,” said Tamara Syrek Jensen, director of the coverage and analysis group at CMS. “So what is the right time for us to get involved?” She noted if a company’s clinical trial is complete, CMS’ involvement could come too late. With Exact Sciences, CMS was in a listening mode for the early meetings with FDA, said the company’s chairman and CEO, Kevin Conroy. Syrek Jensen agreed, saying CMS realizes FDA approval must come first, but CMS had a dialogue with the company outside meetings via email or in phone conversations. FDA approved Exact Sciences’ Cologuard, a stool-based colorectal cancer screening test, in August. The same day CMS issued a proposed national coverage determination that is expected to be finalized in early November. The approval marked the first through the parallel review program and few other companies have publicly expressed interest in the program. Conroy said Exact Sciences is meeting with lawmakers to educate them about the program in a bid to ensure its survival. “We don’t know if this program is going to continue to be funded and we want to build support from an industry standpoint,” he said. He told FDA Week that the company has highlighted how the program helped the company get to market faster and save money in the process. “I’m able to tell the story,” he said. Covidien has put one technology forward to be considered for the program, said Michael Tarnoff, the company’s corporate chief medical officer. He cited government resource constraints as one of the bigger limiting factors for the program. However, for-profit companies want to get their products to market sooner and will prioritize resources for the engagement. “When FDA and CMS invited us to come with our technology, I got six or seven people together and we went in two days,” he said. “So we will resource this and make it happen.” He noted the program has other limiting factors, such as fear of engaging with the agencies. However, he noted that collaboration with the agencies is at an “all-time high.” “There’s a cultural shift underway and I think the times are forcing it but the companies are becoming more aware of what they need to be and these government agencies are becoming more aware that they can’t be unapproachable and adversarial,” Tarnoff said. “They have to be collaborative.” — Alaina Busch McBournie MedPAC Looks To Let Hospitals Steer Patients To PAC Providers Congressional Medicare advisers hope to come up with proposals that would give hospitals more flexibility in steering fee-for-service Medicare beneficiaries to preferred high-quality post-acute care sites, after raising concerns Friday (Oct. 10) with private insurers’ initiatives to manage post-acute care. The Medicare Payment Advisory Commission rejected private insurer models used in Medicare Advantage, such as post-acute care benefits managers or beneficiary incentives for using preferred networks, because of concerns these initiatives wouldn’t work within the fee-forservice framework. MedPAC chair Glenn Hackbarth said the commission would like to offer recommendations on managing post-acute care in the near future, but was unclear as to when that would be. Medicare fee-for-service currently prevents hospitals from recommending particular post-acute care providers to beneficiaries. Commissioners decided to explore finding a middle ground for hospitals between soft steering to preferred post-acute providers and hard steering — pushing beneficiaries to use a particular provider — by recommending tweaks to current Medicare rules. One of the biggest problems with hard steering, commissioners noted, is many hospitals also own skilled nursing facilities and other post-acute care facilities. To inform the discussion commission researchers detailed the ways Medicare Advantage plans and private plans have INSIDE CMS — www.InsideHealthPolicy.com — October 16, 2014 5 moved to manage post-acute care to reduce hospital readmissions and improve patient experience. Researchers said many plans are beginning to contract with post-acute care benefits managers who direct the patients’ medical steps after a hospital stay. But commissioners were reluctant to explore this path because they didn’t want to add a new entity into feefor-service Medicare. Many Medicare Advantage and private plans also offer incentives to use preferred networks of post-acute care settings with beneficiaries being penalized for seeking care outside of the network, commission researchers said. Commissioners said incentivizing patient behavior in fee-for-service Medicare is difficult because many of them have Medigap or Retiree Supplemental insurance that covers copays and co-insurance leaving beneficiaries paying virtually nothing for their care. Additionally the very nature of fee-for-service with its emphasis on beneficiary choice in providers makes setting up post-acute preferred networks difficult, commissioners said. Hackbarth pointed out that preferred networks work in Medicare Advantage, because in that program beneficiaries willingly forgo the open choice of the fee-for-service program. While current Medicare fee-for-service rules state that hospitals can’t steer beneficiaries to post-acute care providers, commission researchers said they believed that hospital physicians and staff, in providing information to fee-for-service beneficiaries on the range of post-acute care providers they can choose from, are already doing some type of soft steering to higher quality post-acute care providers by putting those providers on the top section of lists given to patients or through informal conversations on their personal experience with different providers. As commissioners zeroed in on relaxing rules for hospitals in this regard one commissioner suggested that if hospitals offered fee-for-service beneficiaries two or more options for post-acute care providers that may still qualify as choice. “Hard steering, I’m thinking, could mean a choice of like two or three preferred providers,” commissioner Kathy Buto said. “It wouldn’t need to be this is where you’re going — end of discussion. I think that could make a difference.” Commissioner Warner Thomas, of Ochsner Health System, agreed with Buto, saying the approach would be narrowing choice rather than eliminating choice. Commissioner Scott Armstrong, of Group Health Cooperative, said offering a choice of providers may be “overrated” when the alternative is better care and lower cost. Vice Chair Jon Christianson, of the University of Minnesota School of Public Health, said going forward the commission should focus on recommending changes within the current law that allow hospitals to be more aggressive in recommending post-acute care providers to beneficiaries, which was the consensus of the commission. — Todd Allen Wilson CMS: People With Plans Canceled In 2014 Need Not Re-Apply For Exemption The administration in guidance issued on Monday revealed that people who were allowed to enroll in catastrophic plans because an issuer had canceled their non-ACA compliant policy for 2014 and they were unable to find affordable coverage may now renew that coverage through 2016 without having to re-apply for a hardship exemption. However, the guidance notes that the majority of hardship exemptions obtained for other reasons will be expiring as of Dec. 31. The information comes as part of a guidance to issuers on how to renew coverage for individuals who had been enrolled in catastrophic plans. Catastrophic plans offer less-generous coverage, and therefore less-expensive premiums, than the “metal tier” plans sold through the exchanges. They are available for purchase by consumers under age 30, or by anyone who received a hardship exemption whether due to plan cancellations or other reasons established by HHS. In addition to being allowed to enroll in a catastrophic plan, a hardship exemption also means that individual is not required to pay a penalty for failing to have coverage. As the guidance explains, the administration had created the plan cancellation hardship exemption on Dec. 19, 2013 as part of its response to the outcry over the cancellation of non-ACA compliant insurance plans. The administration had also created a “transitional policy” under which issuers could continue to offer the non-compliant plans for one year if allowed by the state insurance department. The insurance industry had blasted the administration’s decision to allow individuals to purchase catastrophic plans, saying that the move could destabilize the market. “This latest rule change could cause significant instability in the marketplace and lead to further confusion and disruption for consumers,” Karen Ignagni, CEO of America’s Health Insurance Plans, said at the time. A industry source says issuers are still reviewing the latest guidance. In March, the administration extended that transitional policy, allowing people to re-enroll in non-compliant individual and small group plans through Oct. 1, 2016 — which means that those plans could stay in effect until fall of 2017. The administration also said that it would extend the hardship exemption policy for the same time period. Monday’s guidance reiterates that policy, and explains that those originally exempted need not re-apply through 2016. As of June, 38 states had said they would allow the non-complaint plans to stay on the market, although not all states planned to allow the policies to remain through 2016, according to researchers from Georgetown University. Regarding the hardship exemption, David Cusana, senior fellow at the Georgetown Health Policy Institute, says that 6 INSIDE CMS — www.InsideHealthPolicy.com — October 16, 2014 rather than enrolling in a catastrophic plan, a consumer may be better off purchasing a health plan with a higher actuarial value. Most catastrophic plans have an actuarial value of about 57 percent, which is just shy of the Bronze plans’ 60 percent actuarial value, he points out. (Consumers purchasing the metal tier plans through the exchanges have access to the ACA’s subsides. Those buying catastrophic plans do not.) Health expert Tim Jost of Washington and Lee University also suggested that consumers may be better off enrolling in Bronze plans because the catastrophic plans are not that much less expensive. Jost, who is also a consumer representative to the National Association of Insurance Commissioners (NAIC), said that the idea of extending the non-compliant plans was “unfortunate” from the perspective of getting people into the risk pool and driving down costs. Many people who had the non-complaint coverage were lower risk, he points out. Jost also found it disappointing that the administration granted the hardship exemptions to those with canceled plans — and will allow the exemptions to continue — rather than encouraging those individuals to look for coverage. At the very least, the administration should require that those consumers prove they are unable to find coverage that is affordable, he says. Still, Jost says that there is probably only a small number of people who will claim this exemption, so while it’s “questionable as a policy” it is not likely to have a large impact on the risk pools. For the 2014 plan year, 2 percent of the total 8.019 million exchange shoppers purchased a catastrophic plan, with figures ranging from 0 percent in several states to a high of 4 percent in Kansas, according to HHS’ May enrollment report. But the report does not explain the characteristics of those shoppers, so it’s unclear if they were under 30 or had an exemption. Meanwhile, is it expected that the number of people to receive plan cancellation notices this year will be far fewer than last year. The Morning Consult recently reported that about 50,000 consumers will see individual plan cancellations whereas estimates from last fall ranged from hundreds of thousands to millions A March Health Affairs report by Urban Institute researchers found that about 18.6 percent of respondents to the Health Reform Monitoring Survey (HRMS) said that they had received a cancellation notice. Therefore, using the National Institute of Health estimates that about 14 million people had individual plans, the authors conclude that about 2.6 million people had their plans canceled last cycle. — Amy Lotven Hillary Clinton: Device Industry Has ‘Argument To Make’ For Tax Repeal CHICAGO —Hillary Clinton, who is widely viewed as a frontrunner Democratic presidential candidate, told the device industry that it should make its case for the device tax repeal within a larger set of provisions that could be changed in the Affordable Care Act. The former senator and Secretary of State ultimately did not commit support to the device tax’s repeal, a top priority for the industry, but did not rule it out. Clinton — speaking at a keynote speech at the Advanced Medical Technology Association’s medtech conference this week — also acknowledged industry’s concerns about the FDA approval process, but pegged it as a resource issue. She said agencies are under-resourced and strained by sequestration. She highlighted that there are more consumers in the marketplace using medical devices as a result of health reform. Clinton also cited industry’s role in providing medically sound and cost-effective care under a law that she described as promoting innovation and emphasizing the quality of medical care. “And yes I know that you have important questions that you would like addressed, but my view is that we need to keep working toward win-win solutions,” she said. “Improving what we have in sensible ways that will lead to lower costs, greater insurance at affordable costs for everyone, higher transparency for the consumers, who after all bear more and more of the burdens of out-of-pocket costs.” She said Medicaid expansion, if embraced by more states, could have furthered the goal of cutting healthcare costs. Clinton’s comments come as Evan Bayh, former Democratic Indiana senator and governor, earlier in the week told conference attendees that the chances of a device tax repeal are “very good” should the Republicans take the Senate in the November elections. A Republican-controlled Congress likely would first vote to repeal the ACA, and after that measure is vetoed would settle on other changes to the law, such as a repeal of the device excise tax, said Bayh, who is now a partner at McGuireWoods. Bayh said the president is likely to support the device tax repeal so long as it is paid for. Clinton said it is important to keep an eye on costs as stakeholders evaluate what has worked and what could work better in the initial period of ACA implementation. She echoed that any action on the tax would have to fit into broader ACA changes. “I think you have an argument to make,” she said. “I think it has to be made within the context of the larger set of issues that have been raised by the ongoing implementation of the Affordable Care Act.” The 2.3 percent excise tax was levied on medical device firms starting in January 2013 and some companies have been curbing new hires and research and development budgets as a result. “I don’t know what the right answer about the tax is but I think we could — taking a look at everything and not standing there with our arms folded staring at each other across the partisan divide — begin to sort it out,” Clinton said. — Alaina Busch McBournie INSIDE CMS — www.InsideHealthPolicy.com — October 16, 2014 7 CMS Releases Star Ratings For MA, Rx Plans CMS on Friday (Oct 10) released 2015 star ratings for Medicare Advantage and stand-alone drug plans and published plan ratings on Medicare Plan Finder to help seniors choose plans. About 60 percent of beneficiaries who choose Medicare Advantage enrolled in high-performance plans for 2015, CMS says, and 53 percent of Part D benficiaries enrolled in stand-alone prescription drug plans with at least a four-star rating on a five-star scale. In 2009, just 17 percent of Medicare Advantage enrollees and 16 percent of Part D enrollees were in high-performance plans. CMS Administrator Marilyn Tavenner announced in September that she expected high-performance plan enrollment to increase yet again this year. The agency has been pushing plans to increase their quality scores for years with a series of carrots and sticks, and those incentives also steer beneficiaries toward plans with four and five-star ratings. However, plans face new financial pressures next year when they will lose bonuses and face further pay cuts. However, CMS is delaying for one year terminations of Medicare Advantage and prescription drug plans with persistent low performance ratings because it worries that the Star Ratings Program disadvantages plans with unusually large numbers of poor enrollees. The Star Ratings Program calls for terminating plans that score lower than three out of five stars three years in a row and awarding bonuses to plans with high ratings. However, plans that enroll higher numbers of low-income beneficiaries have for a long time complained that the program’s quality measures disadvantage them. The average MA premium submitted by health plans for 2015 will increase by $2.94 next year, according to a CMS release, to $33.90 per month. However, CMS estimates the actual 2015 MA average premium will increase by only $1.30 because more beneficiaries are expected to enroll in lower-cost plans. Separately, CMS announced Thursday that Part B premiums for most seniors in Medicare fee-for-service will remain unchanged for a second consecutive year.—John Wilkerson Coalition Seeks Leniency On Employee Tax Credit Repayments A coalition representing the retail, restaurant, insurance, temporary staffing and other industries is asking the administration to consider a grace period for employees who could face significant repayments of ACA tax credits due to marketplace inaccuracies that improperly awarded them credits for exchange coverage even though they had access to employer-sponsored coverage. The coalition continues to urge administration officials to consider implementing a “prospective” reporting system that could mitigate confusion and protect employees from tax consequences. The request came as part of a six-page letter from the Employers for Flexibility in Healthcare to HHS Secretary Sylvia Burwell and Treasury Secretary Jack Lew outlining a series of outstanding concerns over the ACA’s employer reporting process, notification process, employee eligibility determinations and credit repayments. The letter points out that in some cases employees may try to sign up for subsidized coverage through the exchanges even though they have an offer from their employer that meets the ACA’s affordability and minimum value standards. The law requires employers with 50 or more workers to offer coverage that has a minimum actuarial value of at least 60 percent, and the coverage must cost no more than 9.5 percent of an employee’s household income. Non-compliant employers will be penalized $2,000 per employee for not offering coverage, and $3,000 for each employee that gets a tax credit if the coverage offer is deemed not affordable. In 2014, the group says, there were instances where an employee was offered employer coverage that met ACA standards but declined it and instead sought coverage through the exchange. In some cases, exchanges deemed individuals eligible for credits without verifying whether they had access to employer-sponsored coverage, the coalition says. This raises the possibility that the individual could have to repay all or part of the credit during the tax reconciliation process, the letter adds. Under the law, an employer is only penalized for not providing minimal coverage if the employee receives a tax credit. The law also requires exchanges to notify employers if one of their employees seeks coverage and is deemed eligible for a tax credit, and the employer has the option of responding with information showing that the offered coverage met the ACA standards. HHS has said that employers have not been given the notices because they will not face penalties under the employer mandate for 2014, the coalition says. The mandate was originally slated to go into effect in 2014, but now it will be implemented in 2015 for employers with more than 100 workers, and in 2016 for employers with between 50 and 100 employees. The final rule on the mandate also phased in the coverage requirement, and now businesses with 100 or more employees must provide coverage to at least 70 percent of the workforce in 2015 and to 95 percent of the workforce in 2016 and beyond. Still, coalition members say they are very concerned that their employees could be vulnerable to re-paying the subsidies. The group also worries that the problem could be repeated during plan renewals since that process does not include a 8 INSIDE CMS — www.InsideHealthPolicy.com — October 16, 2014 timely verification process. The coalition raises several concerns about the employer notification process. First, the group points out that HHS has yet to inform employers what type of information will be included in the notice. In 2014, the group says, some states provided notices that were “of little or no use” to employers because they did not include information on which employee had sought the coverage. The group also notes that HHS plans to send notices to the address that the employee reports on the application, which may be an individual store and not the corporate headquarters or other entity that deals with issues related to employee benefits. HHS also has not provided details on the appeals process. The coalition says that HHS is apparently planning to send the notices “in batches beginning this Spring.” This plan, the group says, could result in an employee remaining in coverage that he or she is not eligible for far longer than if there were a more timely response. For example, if an ineligible employee applies for and gets subsidized exchange coverage effective in January, but the employer is not informed until March, that employee could be on the hook for five months of tax credits once the employer’s 90-day appeals process is completed. Additionally, the employee would be ineligible to sign up for the employer coverage at that time, and would thus be stuck either paying the full price for exchange coverage or remaining uninsured. Due to these concerns, the coalition says that employees should have leniency in the repayment process for year one. “In recognition that 2015 is largely a practice year in which exchanges and employers will be testing out and gaining valuable experience with a complex notification and appeals process, we urge to the administration to consider a grace period if such tax consequences occur as a result of inaccurate APTC determinations, delayed notifications, wrong addresses, lack of personally-identifiable information on notifications, appeals backlogs or other process inefficiencies during this learning period,” the group writes. The coalition writes that its members will face “significant challenges” in the coming months as they prepare for the employer mandate to kick in. The letter notes the short time for complying with the law’s requirement that employers report to the IRS detailed information on the coverage offered to their employees is especially problematic. The data is slated to be reported by January 2016, but must be collected starting Jan. 1, 2015, and many business will have to build entirely new systems, the letter says. While IRS released the final employer mandate rule in March, the agency did not release draft forms until July 24 and draft instruction until Aug. 28, and still has not released the technical specifications needed to build the IT systems, the group says. Given the complexity of the changes, the group writes, many business are worried that they will need to collect data manually during the first year. Employers still seek information on when the IRS will release the technical specifications, when the final forms and instructions will be released, how the IRS is communicating that employers must begin the reporting requirements, and whether the IRS will consider the short time frame when it does its economic impact analysis, the letter says. “Retailers have worked tirelessly with the administration on solutions to address the challenges this law presents for employers,” said Christine Pollack, vice president of government affairs at the Retail Industry Leaders Association. “With less than three months before the employer mandate goes into effect, significant issues and pertinent questions remain. Employers are working to comply with the law and it is important that the Administration do all they can to mitigate the upcoming challenges for businesses, employees, Exchanges and the IRS.” The coalition also repeats its call for the administration to create a process for employers to voluntarily report to the IRS that they are offering coverage that meets the ACA requirements. Creating such a system would improve the accuracy of the eligibility determinations and also protect employees from potentially having to repay tax credits, says the group. The coalition has been pushing prospective reporting as a solution for years and argues that the administration has the authority to establish such a system without additional legislation. The group has held numerous meetings with IRS and HHS on the issue but the administration has refused to take action, which is why the coalition is now making a stronger push on Capitol Hill, says a source. Coalition members support legislation offered by Sen. Mark Warner (D-VA) that would required the Treasury to report to Congress on the processes necessary to develop such a voluntary reporting system. That bill now has 10 Democrat co-sponsors including Sens. Mark Begich (AK), Michael Bennet (CO), Al Franken (MN), Heidi Heitkamp (ND), Tim Kaine (VA), Amy Klobuchar (MN), Jeanne Shaheen (NH), Tom Udall (NM) and Joe Donnelly (IN). The prospective reporting provision is also part of Rep. Diane Black’s (R-TN) Streamlining Verification For Americans Act (HR 5557), which would reduce some of the reporting requirements, delay the flow of subsidies to individuals until eligibility can be manually or electronically verified, and require the Comptroller General to evaluate HHS’ progress on the employer notifications. — Amy Lotven INSIDE CMS — www.InsideHealthPolicy.com — October 16, 2014 9 CMS FAQ Provides Certainty For Medicaid Plans . . . begins on page one health insurance provider fee. “The fee is not unlike other taxes and fees that actuaries regularly reflect in developing capitation rates as part of the non-benefit portion of the rate. CMS believes that the Health Insurance Providers Fee is therefore a reasonable business cost to health plans that is appropriate for consideration as part of the non-benefit component of the rate, just as are other taxes and fees,” the agency says. The health insurer fee is expected to raise close to $100 billion from all insurers under the ACA, and MHPA said in a September letter to CMS Medicaid chief Cindy Mann that “significant confusion persists among states regarding the treatment of taxes and fees, including the health insurer fee.” The group had been pushing CMS for months to clearly lay out guidance on how states should treat the health insurer fee for managed Medicaid plans. CMS says the FAQ is the result of a number of questions from states, their actuaries and health plans about the fee and its interaction with managed care rate setting. Myers said there are states that still haven’t contracted with plans to pay for the fee, and the FAQ provides certainty on that front. The JP Morgan note on the FAQ says that CMS’ move increases the likelihood that the remaining states that haven’t yet committed to paying the fee will do so. “However, the FAQ appears to still leave some wiggle room for states on grossing up reimbursement for income taxes, so the uncertainty has not been eliminated completely,” the JP Morgan note states. In response to a question on whether the potential effect of the tax on other fees as well as the non-deductibility of the fee should be considered in the capitated rates, CMS says only that “it may be” factored into rate development. If so, the actuary will include that data in the underlying assumptions, the agency says. Medicaid plans have argued that all impacts of the fee should be reimbursed under actuarial soundness principles. The JP Morgan investors note says that Texas is the key hold out on covering the fee, as the CEO of the Texas Health Plan Association sent an email late last month saying it didn’t expect the Texas Health and Human Services Commission to do anything about the fee soon. The email also said that HHSC wasn’t making changes to indicate that the state should pay the insurer fees, the investors note says. “We expect the state to address the fee in the 2015 budget process, with the biggest uncertainty at this point being whether or not the state decides to retroactively reimburse the plans for 2014,” the note says. In the FAQ, CMS says states have flexibility in incorporating the health insurance provider fee into the states’ managed care capitation rates, and can account for the fee on a prospective or a retroactive basis. It’s tough for an actuary to make a judgment about what the fee will look like a year ahead of time, so CMS did the right thing in granting that flexibility, Myers said. CMS says it expects that states will move to a prospective calculation as states and the plans get a little more experience with the fee. If a prospective calculation ends up making the capitation rates too high or too low, then the rates may be adjusted after the tax assessment is known, CMS says. States can also account for the fee by withholding some funds until a health plan’s actual fee is known, the FAQ adds. The agency says that the health insurance providers fee cannot be paid to the plans as a separate payment, and “any payment for the fee — whether on a prospective or retrospective basis — must be incorporated in the health plan capitation rates and reflected in the payment term under the contract.” CMS says that it’s reasonable to consider the fee in risk-sharing and minimum medical loss ratio calculations, since they may exist in the contract between the state and the plan. The FAQ also says that if a plan contracted with a state to provide managed care for the Medicaid population in 2013 SUBSCRIPTION ORDER FORM Sign me up to receive Inside CMS at $705 per year in the U.S. and Canada; $755 per year elsewhere (air mail). Name __________________________________________________ Affiliation _______________________________________________ Address _________________________________________________ City/State/Zip ____________________________________________ Phone __________________________________________________ E-mail __________________________________________________ To order by mail: Send this coupon to Inside CMS, P.O. Box 7167, Ben Franklin Station, Washington, DC 20044. To order by phone, fax or e-mail: Call 800-424-9068 (in the Washington, DC area, 703-416-8500). Fax to 703-416-8543. E-mail at [email protected] Please check one: Visa MasterCard Bill me Check enclosed (DC subscribers add 6% sales tax) Card number _________________________________________ Name on the card _____________________________________ Signature _______________________________________________ 10 Exp. date ____________________________________________ INSIDE CMS — www.InsideHealthPolicy.com — October 16, 2014 but not 2014, then the process for reimbursing a health plan for the fee is a matter to be negotiated between the state and health plan. But any retroactive rate adjustments for a plan that left the market must be made under the contract and within the federal two-year period for timely claims, CMS says. The agency also says that states that choose to reimburse health plans for the health insurance provider fee after the amount is known should have a timeline for payments. Typically, this means the states should reimburse plans between 30 to 90 days after the amount of the fee is known, the FAQ says. Myers said plans are excited about this, as they beleive they should be paid in a timely fashion. CMS recently released a 2015 Managed Care Rate Setting Consultation Guide, as well, which said that taxes, fees and assessments data should be included as part of the projected non-benefit costs actuaries include when developing the managed care rates for 2015 and showing they are actuarially sound. Both Myers and the JP Morgan investors note say this indicates more rigorous CMS oversight of Medicaid rate setting. The GAO in 2010 said that CMS’ oversight of states’ rate setting needed improvement, and MHPA has been pushing for more transparency in the rate setting process. The agency says that “[t]he actuarial review of the rates and the use of the 2014 Consultation Guide have increase the transparency of the rate development process and have led to a better understanding of expectations between states and CMS on Medicaid managed care rate setting and CMS’ oversight of the process.” Most of what CMS lays out is already part of the states’ actuarial work and program management, the agency says. The guide points out that the capitation rates for the new adult population may vary from the rates for the traditional Medicaid population, but “those reasons must be documented and justified in the certification,” and not occur because of a difference in the amount of federal assistance for the different populations. — Michelle M. Stein OIG Pushes To Equalize Cost-Sharing At CAHs, Other Hospitals Medicare beneficiaries pay nearly half of the costs of outpatient services at critical access hospitals, which is significantly more than those beneficiaries would have paid if they accessed the same care at other hospitals, the HHS Office of Inspector General says in a recent report that has sparked debate on how to fix the problem. The National Rural Health Association calls the difference in beneficiary’s cost sharing an unintended consequence of the CAH’s reimbursement system, and says that since CMS and Congress created this problem they should be the ones to fix it. Both the OIG and the NRHA note that if lawmakers were to equalize beneficiaries’ cost sharing between hospitals paid through the outpatient prospective payment system and the CAH reimbursement system, Medicare would need to pay more. The OIG suggests that Medicare could limit increases in the program’s cost by cutting back on the number of CAHs through looking again at distance requirements — an approach blasted by hospitals in the past. The OIG recommends that CMS ask Congress for authority to change how the CAHs are paid in order to address the problem. The OIG also suggests CMS could calculate beneficiaries’ coinsurance off of a CAH’s interim payment rates, rather than the hospital’s charges. Another option the OIG suggests is processing claims for CAHs the same way that a claim at non-critical access hospital would be processed under the OPPS in order to calculate what the beneficiary owes. CMS would then pay the rest of the CAHs’ reasonable costs up to 101 percent. The OIG notes that Congress’ Medicare payment advisers (MedPAC) provided CMS with options on how to fix the situation as part of a 2011 report. The discrepancy in cost sharing stems from the fact that while most hospitals are paid through the OPPS system, CAHs are paid differently and receive 101 percent of their reasonable costs. Beneficiaries pay 20 percent of the hospitals’ charges — which are usually higher than the CAHs’ reasonable costs — and then Medicare will pay whatever else a CAH is owed. This means beneficiaries paid nearly half of the costs for outpatient services at CAHs in 2012, the OIG says. Beneficiaries paid about $1.5 billion of the estimated $3.2 billion in outpatient CAH costs, according to the report. For one of the outpatient services the OIG looked at, beneficiaries’ average coinsurance at CAHs was more than the total OPPS rate. The average percentage of costs that beneficiaries paid to CAHs also increased between 2009 and 2012, the report found. The NRHA says in a blog post that “the current CMS policy unfairly penalizes rural patients, which is not the intent of Congress,” though the group also notes that the hospitals are simply billing the beneficiaries according to the rules set up by Congress and CMS. “We ask Congress to direct CMS to not shift the burden to providers or to patients,” the NRHA says. “This problem was created by Congress and Medicare and can be fixed by them with ease. As the OIG report documents the problem, it’s time to fix this inequity,” the group adds. NRHA suggests that Medicare could easily fix the unintended consequences of the CAHs’ reimbursement system by holding the beneficiaries harmless on coinsurance amounts between the CAHs and hospitals under the INSIDE CMS — www.InsideHealthPolicy.com — October 16, 2014 11 prospective payment system. Both NRHA and OIG note that lowering how much beneficiaries pay means that Medicare’s costs will go up. If CMS wanted to limit the increase in Medicare costs, the OIG suggests, the agency could phase in any changes over several years. The agency could also “offset the potential increase in Medicare expenditures by ensuring that only those CAHs that meet all participation requirements continue to receive cost-based reimbursement,” the report says. The OIG previously looked at distance requirements between CAHs, and found that nearly two-thirds of the CAHs are too close together to meet the location requirements. The American Hospital Association and NRHA blasted the report, and said decertifying critical access hospitals that didn’t meet the location requirement would endanger the rural health system. NRHA disagrees with the OIG’s suggestion that CAHs contribute to fixing the problem around beneficiary cost sharing through a CMS recertification of CAHs and a determination of whether they meet the location requirements. — Michelle M. Stein Health Plans Argue Harvoni Price Too High . . . begins on page one letter to Congress complaining that Gilead didn’t take advantage of a “golden opportunity” to respond to criticism from patients, policymakers and payers about Sovaldi’s $84,000 treatment regime cost. “Instead, Gilead chose to double-down with a price of $94,500 for Harvoni,” he wrote. Rother urged lawmakers to force drug makers to justify and document their pricing decisions, unveil projected prices earlier in the FDA approval process, and make more clinical safety and efficacy data available to researchers. Medicaid Health Plans of America timed the release of a report on states’ responses to Sovaldi’s price with Gilead’s Harvoni announcement. The report, prepared by Viohl & Associates, warns the growth of specialty pharmacy drugs will hike pressure on states to manage treatment costs. “States will continue to employ a variety of strategies and impose restrictions, including prior authorization and quantity prescribing limitations, to curtail the impact of such medications on their Medicaid programs,” the report concludes. While Gilead’s $94,500 price for a 12-week course of Harvoni is less than some market analysts expected, Rother complained the company merely set the price to be on par with the cost of a treatment course of Sovaldi, plus pegylated interferon and ribavirin. The price is not based with any regard to “value proposition or cost/benefit calculations,” he wrote to lawmakers. Insurance companies and others have been pressuring Gilead to lower the Sovaldi’s price and had hoped the company would set a new course with its successor drug Harvoni. Harvoni is a once-daily pill that combines Sovaldi (sofosbuvir) and ledipasvir. A Gilead spokesperson said the price of Harvoni reflects the value of the medicine because it cures patients for significantly less than the cost of treating patients indefinitely for the chronic disease. The price per bottle for Harvoni is $31,500. Hepatitis C patients without scarring of the liver and with low viral loads can be cured with an eight-week course, Gilead says, and those patients account for about half of those infected with the most common type of hepatitis C. Given that the price per-pill is the same regardless of the length of the treatment course, the eight-week course costs $63,000 and the “average blended” cost of the drug is $81,000. But Rother told Inside Health Policy that most plans will only cover the drug for sicker patients so he doesn’t buy the so-called blended cost. Also, the market will be expanded because Harvoni eliminates the need to take interferon, which makes patients very sick. “In the rest of the economy, when you get a bigger market, you cut the price for volume,” Rother said. Some analysts expected Harvoni’s price to be higher because doctors often prescribe Sovaldi with Johnson & Johnson’s Olysio, even though FDA did not approve the drugs in combination, and the drugs together cost about $150,000. However, Gilead President and CEO John Milligan told Inside Health Policy on Oct. 1 that Gilead would keep the price close to the price of Sovaldi plus interferon and ribavirin. But health plans complain that the price is too high because some 3.2 million Americans are estimated to be infected with hepatitis C. Other drugs, especially biologics that treat cancer, are more expensive, but they treat much smaller populations. State Medicaid directors and the private managed care plans that administer most Medicaid programs are especially upset. The study released by Medicaid Health Plans of America notes how difficult it is for states to cover Sovaldi without cutting other health services or dipping into education and infrastructure budgets. The analysis finds that 35 states require prior authorization for Sovaldi. Most, if not all, states with prior authorization requirements require liver biopsies to determine the severity of disease before covering the drug. Many states include additional restrictions, including limits on who may prescribe and requirements that patients not use alcohol or illegal drugs. Patient advocates are working with researchers at Harvard and Brown to analyze state restrictions on Sovaldi, but 12 INSIDE CMS — www.InsideHealthPolicy.com — October 16, 2014 unlike health plans, patient advocate are doing the research to advocate for lifting restrictions, especially those that prohibit alcoholics and patients addicted to illegal drugs from getting Sovaldi. The analysis by Medicaid plans finds that, of the 37 states that use managed care plans, 28 include pharmacy benefits and nine states carve out the pharmacy benefit. Also, 22 states do not require that Medicaid managed care plans align preferred drug lists with state-approved lists. — John Wilkerson INN experts meet... WHO’s Biologic Naming Plan Draws Questions of Retroactive Naming, Use Key biosimilar and biologic developers came down on different sides as to how a World Health Organization plan for biological qualifiers should apply in practice, with Hospira pressing for its use only for pharmacogivilance, not for prescribing, and originator companies pushing to have BQs appear wherever International Nonproprietary Names are used, including prescribing. The use of BQs and other key outstanding issues, such as how biologics could be retroactively named, are likely to be discussed at the WHO’s next naming meeting, which starts Tuesday (Oct. 14). The issue of whether to name biosimilars differently than their originator products has been one of the most controversial issues surrounding FDA’s implementation of the health law’s biosimilar pathway, and WHO’s plan has fueled the debate. WHO in July laid out a plan for biological qualifiers — an alphabetic code added to the INN. This week the international organization will hold its first meeting since formally unveiling the plan, with the 59th INN consultation starting Tuesday. In response to the plan, various stakeholders opposed linking the biological qualifier to manufacturing sites, with some citing that as their only major concern. Many support the fact that WHO’s plan creates a qualifier that is separate from the INN and would apply to all biologics. Sources said one component that has yet to be worked out is whether and how existing biologics would be renamed under a system that applies to all biologics. “I think that’s had a lot of people scratching their heads,” said Ginny Acha, Amgen’s director of regulatory policy in Europe. She noted that was one area where the WHO plan was “light” on details. WHO will also have to get buy-in from international regulators as it cannot mandate a naming plan. Acha said she doesn’t “underestimate the challenge” and building global support for the BQ approach will require educational efforts, workshops and feedback from regulators. “The way they’ve done it with INNs is they’ve proven the value of it,” she said. Amgen, which develops biosimilars and originator biologics, supports distinguishable naming and the “overall direction” of WHO’s policy on naming. “Amgen believes that healthcare systems globally must ensure all biologic medicines, including biosimilars, can be rapidly and accurately identified by national regulators, healthcare providers and patients,” according to the company’s policy statement. “Distinguishable naming conventions for all biologics will help strengthen the accuracy of tracing via post-marketing safety monitoring systems.” In addition to the potential for retroactive naming, questions remain about how BQs would be used throughout the healthcare system. The WHO plan provides a list of potential uses, such as for reimbursement, prescribing and substitution. The Biotechnology Industry Organization and European industry groups ask that the BQs be used alongside the INN. “[F]rom a functional perspective, BIO strongly encourages WHO to clarify in the final policy that the BQ scheme will work only if the BQ is used in all circumstances where the INN is used, including labeling, prescribing, advertising and promotion, adverse event reporting, record keeping, etc,” the industry group tells WHO. “BIO believes this should be facilitated by strongly encouraging national/regional regulatory authorities to include the BQ as part of the local nonproprietary name (i.e., for the United States, the United States Adopted Name or USAN).” European Biopharmaceutical Enterprises (EBE) Association and the European Federation of Pharmaceutical Industries and Associations (EFPIA) made a similar argument. “If the BQ is not part of the complete name, then it is less likely to be captured accurately in (pharmacovigilance) systems and products will not be identified accurately,” the groups tell WHO in comments. But Hospira is pressing for a different approach and opposes using BQs for prescribing. “You can’t ask a physician to memorize 20 qualifiers,” said Julie Reed, Hospira’s vice president of global governmental affairs. She argued that the conversation around BQ uses should be narrowed. Interchangeability, for example, is only part of the biosimilar pathway in the United States, but listed as a possible use for the BQ. Hospira says regulatory bodies in highly developed regulatory environments should continue their existing systems, but the BQ approach could be used in markets that have less-developed biologic and biosimilar regulations. The company supports the WHO proposal with some caveats. “First, Hospira vigorously opposes the suggestion that the qualifier could or should be used by physicians for INSIDE CMS — www.InsideHealthPolicy.com — October 16, 2014 13 prescribing biologics,” Hospira tells WHO in comments. “Our experience in Europe is that physicians prefer prescribing by brand name, and we believe that a system that requires a physician to prescribe using a random code will lead to medication errors and confusion.” The company also says the BQ system should not replace well-functioning pharmacovigilance systems and WHO should delete or modify the list of potential BQ uses. — Alaina Busch McBournie AMA Wants More Flexibility In MU Program . . . begins on page one AMA floats a blueprint for revamping the program that calls for CMS to: ease the thresholds for compliance; get rid of the rigid list of core requirements; expanding opportunities for hardship exemptions; align the Physician Quality Reporting System with meaningful use requirements; add exemptions for hospitalists and physicians close to retirement; craft standards for sharing and transmitting data between EHRs and clinical data registries; ease restrictions on who can enter a physician’s orders; and streamline the MU vendor certification process. In a follow-up to Tuesday’s letter, AMA along American Academy of Family Physicians, Medical Group Management Association, National Rural Health Association and several health care systems signed on to a brief letter Wednesday (Oct. 15) to HHS Secretary Sylvia Mathews Burwell asking that the MU program be streamlined to promote interoperability and the usability of health information technology much in the way AMA laid out in its own letter to CMS. AMA contends that while the meaningful use program is designed to increase efficiency and improve the patient experience, the fast pace and strict timelines imposed are impeding the work flow of physician practices and hospitals and taking time away from patient interactions. Physicians and hospitals, AMA says, are having difficulty in meeting measurement requirements that in some cases are out of date, leaving them facing penalties. This is coupled with a high cost to implement vendor products that are often not fully tested — which leads to problems with interoperability — and is causing negative returns on investment that ultimately disincentive providers from participating in the program, AMA adds. “While more than 78 percent of physicians are using an EHR, thousand have not participated in the Meaningful Use program or attested to Stage 2, in large part because of the program’s all or nothing approach,” said AMA President Robert Wah. “Physicians should not be required to meet measures that are not improving patient care or use systems that are decreasing practice efficiencies. Levying penalties unnecessarily will hinder physicians’ ability to purchase and use new technologies and will hurt their ability to participate in innovative payment and delivery models that could improve the quality of care.” Relaxed penalty and incentive criteria. AMA specifically asks that CMS remove the “all-or-nothing” approach for meeting meaningful use requirements that requires 100 percent compliance to earn in an incentive or face a penalty. The physicians group says CMS should adopt a system that lets doctors earn incentives by meeting 75 percent of compliance requirements and face a penalty if they do not meet a 50 percent threshold. AMA says that in order to make this work CMS should also get rid of the “core” and “menu” requirements for attestation and allow doctors to choose a number of measures from a master list that best reflects the particulars of their individual practices. AMA points out that there is precedence for such an approach under the Medicare e-Prescribing program. Expanded hardship exemptions. The organization also wants CMS to expand the hardship exemptions for the MU program to include an exemption for physicians who successfully participate in Physician Quality Reporting System Program (PQRS). AMA notes that PQRS and MU both require reporting quality measures but have different timelines, which can place a “duplicative burden on physicians.” Alignment of PQRS and MU. The doctors group urges CMS to align the PQRS and MU to make the timelines and quality measures reporting match and ensure a process exists for public input on new electronic clinical quality measures. AMA would also like to see a process developed for getting rid of measures that no longer meet latest clinical practice. More exemptions. The group also urges the agency to keep the exemption for anesthesiologists, radiologists and pathologists going forward, as well as adding exemptions for hospitalists and physicians close to retirement. New data-sharing standards. AMA would like the agency to develop standards for sharing and transmitting data between EHRs and clinical data registries in order to streamline the process and eliminate the current need for often having to manually input data into clinical data registries. Eased limits on physician’s orders. Easing restrictions on who can enter a physician’s orders would make the data entry process more efficient, AMA says, noting that current requirements limiting this entry to physicians and credentialed medical assistants have increased the providers’ administrative duties that now take up to 30 to 40 percent of the work day. To alleviate this problem, AMA says, the agency should allow other trained individuals — such as medical scribes — 14 INSIDE CMS — www.InsideHealthPolicy.com — October 16, 2014 deemed appropriate by the individual provider to input physicians orders into their systems. Streamlined vender certification. AMA also asks CMS to streamline the MU vendor certification process to enable higher-performing products that focus on interoperability, quality measurement reporting and privacy and security to enter the marketplace. The organization would specifically like standards set for vendors for implementing User Centered Design which makes EHR use easier for physicians. — Todd Allen Wilson HHS’ HIT Advisors Say Provider Progress On ICD-10 Prep Slowed By Delay HHS’ health information technology advisors say health plans and vendors are making progress with their ICD-10 preparations, but Congress’ decision to delay implementation of the new code set has posed hurdles because providers have slowed down efforts to prep for the transition or even put them on hold. The advisors recently warned HHS Secretary Sylvia Burwell that there will be a “significant disruption” on Oct. 1, 2015, when the new CMS coding system is slated to go live, unless all parts of the industry make a concerted effort to continue moving forward with ICD-10 preparations. The Workgroup for Electronic Data Interchange completed its ninth ICD-10 readiness survey, and found a split between large providers that had mostly finished their provider impact assessments for ICD-10, and some small providers that are unsure when they will complete these assessments or think they won’t be done until next year. The survey also found that ICD-10 testing has been slowed down or put on hold by a majority of providers as a result of the ICD-10 delay instituted by Congress as part of the latest patch to the flawed Medicare physician payment formula. CMS Administrator Marilyn Tavenner had said in February that there would be no further delays to ICD-10 despite calls to delay its implementation beyond October 2014. “Let’s face it guys, it’s time to move on,” Tavenner said, noting previous delays. But lawmakers pushed the date back to October 2015 shortly thereafter in what some saw as a consolation prize for physicians who did not get a permanent fix to the Sustainable Growth Rate formula. “It appears the delay has negatively impacted provider progress, causing two-thirds of provider respondents to slow down efforts or place them on hold,” WEDI Chair Jim Daley wrote to Burwell. “While the delay provides more time for the transition to ICD-10, many organizations are not taking full advantage of this additional time.” After numerous delays in implementing ICD-10, one health technology lobbyist said, the industry is past the point where more time will lead to better results. Some stakeholders may not make the necessary preparations for ICD-10 until the costs of not switching to the new system become a reality, the lobbyist added. Daley said on a webinar Thursday (Oct. 9) it’s particularly troubling that some providers say they don’t know when an impact assessment for ICD-10 will be completed. Those providers can’t prepare to transition to the new system if they don’t yet know what needs to be done, he said. About three-quarters of small providers said they either didn’t know when they would finish their ICD-10 impact assessment or would complete the assessment in 2015. A lack of progress on which providers have completed, or plan to complete, their impact assessment is a cause for concern, the survey says, as it will leave little time to address any internal issues or testing. Over half of large providers had begun external ICD-10 testing, while most smaller providers didn’t plan to do so until 2015 or didn’t know when they would start testing. A majority of providers said the delay in ICD-10 implementation led them to slow down or put testing plans on hold, the survey says. Venders who were surveyed said the main reason for the lack of progress in ICD-10 planning and implementation are delays in customer readiness. Robert Tennant, a senior advisor with the Medical Group Management Association, said that answer is disconcerting to providers. Providers cannot be ready without the necessary ICD-10 software, so this creates a “chicken and egg situation,” Tennant said. Melanie Endicott with the American Health Information Management Association, which includes vendors, said that most of AHIMA’s members are ready to go. The implementation delay was very frustrating for many members, she said. Tennant said the group is advising its members to take low-cost, high-impact steps to prep for ICD-10 even if their software isn’t ready. There is a lot of uncertainty around whether the Oct. 1, 2015 implementation date will stick after multiple delays, Tennant said, making some leery of spending the time and money to prepare for ICD-10 only to have the implementation date again delayed. If providers choose to hedge their bets, they should do so intelligently, Tennant said. When asked what to do in a situation where preparing for ICD-10 feels like “crying wolf,” Daley said that the industry needs to think about how not being prepared for the transition would affect stakeholders’ reputation, business and operations. Oct. 1, 2015 is the date that has been set, and the industry has to trust as best it can in the government, Daley said. Stakeholders can hedge their bets if they must, but they need to make sure they can move forward to implement the coding system when they need to, he added. The health technology lobbyist said that the industry won’t have a clear picture of who will be prepared and who won’t be ready for the transition until closer to the implementation date. Endicott also said she expects to see a big increase in ICD-10 preparations across the board in 2015. — Michelle M. Stein INSIDE CMS — www.InsideHealthPolicy.com — October 16, 2014 15 PhRMA Says HRSA Misinterprets Statute . . . begins on page one while his court had vacated HRSA’s 2013 legislative rule that said the 340B orphan drug exclusion only applied when a drug was used specifically for its orphaned designation because HRSA didn’t have the authority to issue legislative rules, the agency’s July 2014 interpretive rule in response to the court action was not a part of PhRMA’s original lawsuit. Contreras said PhRMA was well within its rights to initiate a new legal action challenging the July 2014 interpretive rule. The industry group argues in its new legal action that HRSA’s interpretive rule — which is the same policy as the vacated legislative rule — goes against congressional intent and should be vacated. “While we value the hard work and efforts of all agencies, it is important federal agencies recognize and work within the bounds set by Congress. PhRMA is therefore filing suit against the U.S. Department of Health and Human Services to challenge its second attempt to issue a rule conflicting with the plain language of the statute,” PhRMA executive vice president and and general counsel Mit Spears said in a press release. PhRMA sued HHS last fall over HRSA’s regulation that implemented the health law’s orphan drug policy in the 340B drug discount provision which offers discounted pharmaceuticals to safety net hospitals. The Affordable Care Act expanded the 340B discount program to cancer centers, critical access hospitals, rural referral centers and sole community hospitals for outpatient drugs, but the law excluded orphan drugs — drugs used to treat rare diseases or conditions affecting fewer than 200,000 people nationally - from the discount program. In July 2013 HRSA issued a legislative rule that stated facilities with the 340B designation could still get orphan drugs at a discount if they planned to use the drugs to treat conditions that are not part of the orphan indication. Some orphan drugs may have multiple indications or they may be used off label. The court ruled in May that HRSA did not have the authority to issue a legislative rule and vacated the 2013 final rule, but asked PhRMA and HHS to submit supplemental briefs as to whether the legislative rule could survive as an interpretive rule. HHS declined to submit additional briefs, but continued to apply the rule, saying on its website: “The court did not invalidate HRSA’s interpretation of the statute. HHS/HRSA continues to stand by the interpretation described in its published final rule, which allows the 340B covered entities affected by the orphan drug exclusion to purchase orphan drugs at 340B prices when orphan drugs are used for any indication other than treating the rare disease or condition for which the drug received the orphan designation.” HRSA says on its website that the interpretive rule does not create binding norms and requirements; rather it is just the agency’s interpretation of the statute. The agency explains this means that if HRSA takes action against a drug manufacturer or covered entity based on this interpretation, a drug manufacturer or covered entity can legally challenge HRSA’s interpretation of the statute as a defense against the enforcement action. An drug industry lawyer who has been following the 340B orphan drug exclusion legal battles said PhRMA’s new lawsuit will focus on HRSA’s specific interpretation of the statute. The lawyer said however the case is decided it won’t impact HRSA’s overall rulemaking authority as the previous PhRMA lawsuit against HRSA’s 2013 legislative rule did. In Thursday’s filing PhRMA argues that if Congress wanted the 340B orphan drug exclusion to be use-based the lawmakers would have written the statute that way. The exclusion statute specifically states the 340B pricing “shall not include a drug designated by the Secretary under section 526 of the Federal Food, Drug and Cosmetic Act for a rare disease or condition.” PhRMA points out that FDA has said the orphan drug designation applies to the drug itself, “that is to the drug’s specific ‘active moiety’ or molecule.” “Thus, the designation that triggers exclusion under § 256b(e) is not limited to only orphan uses of the orphandesignated drug, as HHS contends. If the drug has an orphan designation, newly eligible entities are not entitled to purchase it at 340B prices regardless of how the drug is used, and must instead purchase the drug at the same price as all other, non-covered entity buyers,” PhRMA writes in its court filing. PhRMA also argues that if Congress had intended the 340B orphan drug exclusion to be “use-based” as opposed to “designation-based” it would have made that clear in the statute as it has done in other instances. PhRMA goes on in the filing to list a number of specific instances when lawmakers have said “only when used for a rare disease or condition;” and “a prescription drug that has been designated as a drug used for a rare disease or condition … unless the human drug application includes an indication for other than a rare disease or condition.” “Any final agency action restricting the scope of that statutory provision, no matter how HHS classifies its action, would be inconsistent with the plain language of the statute and impermissibly create new legal effects on regulated parties that Congress did not enact into law,” PhRMA writes in its court filing. Safety Net Hospitals for Pharmaceutical Access steadfastly disagrees with PhRMA’s interpretation of the statute, saying HRSA’s interpretive rule is well-reasoned and legally valid. SNHPA points out that, according to Magellan Rx Management’s 2013 Medically Pharmacy Trend Report, the drugs Alimta, Gammagard, Herceptin, Orencia and Procirt all have orphan drug designations but are prescribed for non-orphan uses more than 95 percent of the time. “Once again, Big Pharma is trying to increase its prices at the expense of rural and cancer hospitals and their pa16 INSIDE CMS — www.InsideHealthPolicy.com — October 16, 2014 tients,” said SNHPA President and CEO Ted Slafsky. “These providers depend on 340B savings to serve needy patients and, in many cases, to keep their doors open.” The American Hospital Association echoed SNHPA’ comments, saying if the court throw’s out HRSA’s interpretive rule safety net hospitals will suffer. “PhRMA’s unrelenting efforts to argue a narrow interpretation of the Affordable Care Acts (ACA) Orphan Drug Exclusion would nullify the benefits of the 340B program for critical access and sole community hospitals, and rural referral centers supported by the very same law. The 340B program has made it possible for eligible hospitals to give patients better access to the drugs they need. The AHA supports HHS’s effort to extend the benefits of the 340B program to rural communities and the AHA will continue to defend the purpose of the program,” AHA vice president for coverage policy Jeffrey Goldman said in a statement sent to Inside Health Policy. — Todd Allen Wilson Vit als: A Vitals continued on next page Health P olicy Blog Po Excerpts of Inside Health Policy Blogs E&C Announces New General Counsel, Oversight Chief Counsel House Energy & Commerce Chair Fred Upton (R-MI) announced a series of staff changes Friday (Oct. 10), including an incoming general counsel and oversight subcommittee chief council. Karen Christian will take on the role of general counsel with Peter Kielty as deputy general counsel. Christian is vacating her position as chief counsel for the oversight subcommittee, which she has held for the past two years. She has been on the oversight subcommittee staff since 2006, according to a committee statement. Charles Ingebretson will take over as Oversight and Investigations Chief Counsel. Ingebretson was a member of the committee staff in the 1990s and has spent the last several years in the private sector. — Michelle M. Stein Medicare Part B Premiums, Deductible Flat For 2015; Part A Deductible Up Slightly The Medicare Part B monthly premium and annual deductible for 2015 will remain flat at the rates of the last two years, CMS announced Thursday (Oct. 9), though the Part A deductible will go up by $44 for 2015. The Part A (inpatient) deductible will increase to $1,260 — a modest increase, according to CMS. For those in the hospital, the daily coinsurance will be $315 for days 61-90 of hospitalization in a benefit period, $630 for lifetime reserve days; and $157.50 for days 21-100 of extended care services in a nursing home. But premiums for Part A, which are paid by a very small number of beneficiaries, will be cut by $19, falling to $407 per month. The Part B (outpatient) monthly premium will remain at $104.90 for 2015, and the deductible will stay at $147. The higher premiums that seniors with higher incomes must pay will also stay at the same rate as 2014. “This leaves more of seniors’ cost of living adjustment for Social Security in their pockets,” the agency says. Stacy Sanders, the federal policy director at the Medicare Rights Center, says that the flat Part B premium is great news, particularly for low income beneficiaries. It’s also good for beneficiaries to have this information going into open enrollment as they look for plans, Sanders said. Medicare open enrollment began Oct. 15. The Medicare Trustees report had suggested that Part B premiums would be flat, so CMS’ announcement was expected, Sanders said. David Lipschutz, senior policy attorney with the Center for Medicare Advocacy, says the Part B premiums were in line with the trends that have shown low Medicare cost growth over the last few years. Over the past four years, per capita Medicare spending growth averaged 0.8 percent annually, CMS points out in a release on the 2015 Medicare rates. This is much lower than the 3.1 percent annual increase in per capita Gross Domestic Product over the same time, CMS adds. — Michelle M. Stein Groups Lobby For Legislation Clarifying HIT Regulation By Year’s End A coalition of 58 companies, patient groups and health information technology stakeholders this week called on Congress to follow up on an FDA Safety and Innovation Act report and pass legislation that would clarify HIT oversight. The groups contend that HIT could improve the efficiency and quality of health care delivery and regulatory uncertainty stifles innovation. They say there is consensus about the need for a risk-based HIT framework and it’s a bipartisan issue. FDASIA charged FDA and the Office of the National Coordinator for Health Information Technology (ONC) and the Federal Communications Commission (FCC) to come up with a risk-based framework for regulating HIT. Those recommendations were released in April and the agencies held a public meeting in May. The groups want Congress to act on the recommendations. “Before the end of the year, we urge Congress to provide much needed statutory clarity and a stable foundation for continued innovation in health IT,” according to the letter. “This overdue action will allow the Administration to focus its limited resources, staff and expertise on ensuring the safety of new medical technologies that pose the highest potential risk to patients, and will promote a new era of medical innovation that will improve care and lower costs.” Groups signing onto the letter include Health IT Now, IBM, U.S. Chamber of Commerce, athenahealth and Retire Safe. Final FDA guidance says the agency will regulate INSIDE CMS — www.InsideHealthPolicy.com — October 16, 2014 17 medical applications that function similar to medical devices while exercising enforcement discretion on low-risk mhealth applications. — Alaina Busch McBournie Express Scripts: Specialty Drugs Drive Exchanges’ Rx Costs A new Express Scripts analysis of prescription drug costs in the ACA’s exchanges shows that specialty drug costs represent 38 percent of total spending on prescriptions even though those drugs account for just 1.3 percent of pharmacy claims volume, reflecting the high cost of treating the serious illnesses of many new enrollees. Express Scripts says the findings back a need for exchanges to cover clinical drug management. The study concludes that it’s “vitally important” given the increased prevalence of specialty conditions for the plans to provide patients with clinical programs that ensure “appropriate management of complex, high-cost medications,” leading to better outcomes and reduced costs. By contrast just 28 percent of total pharmacy spending in traditional health plans can be attributed to specialty drugs, according to the new report from Express Scripts. Also Express Scripts found that exchange enrollees use more generics, and that silver plans — those with lower premiums but higher out-of-pockets costs for brand-name drugs — have been the most popular among exchange consumers, particularly among those eligible for subsidies. “By preferring generic medications over more expensive brand-name alternatives, these members are using their plans in the way they were designed.” Another finding is that enrollees who signed up on or after March 1 are younger by an average of four years, use fewer medications and have lower incomes than the earlier enrollees. — Christopher Cole Group Urges FDA To Treat Other Experimental Drugs Like Ebola Drugs The Goldwater Institute, the group driving “Right to Try” state laws, said if FDA is willing to forgo its own process when authorizing use for experimental Ebola drugs then it should do so for everyone. The institute is behind “Right to Try” state laws, which have passed in Colorado, Louisiana and Missouri, that would provide patients with experimental drugs earlier than is the case under FDA’s expanded access program. The group in the past has described FDA’s expanded access program as “timeconsuming and extremely difficult to navigate,” according to a February 2014 report on its website. “If these doctors were allowed access to experimental medication without following the FDA’s normal process, we want to know why and how other sick or dying Americans can also get an exception,” said Jon Riches, an attorney at the Goldwater Institute, in a press release. The group requested information on the process used by FDA to authorize use of the experimental Ebola drug, but FDA said it could not release that information because doing so would violate the drug maker’s trade secrets. — Erin Durkin Administration Offers Guidance On Methods For Reference Pricing The three federal agencies charged with implementing the Affordable Care Act on Friday outlined how they will determine whether fully insured or self-funded group health plans that use controversial reference pricing policies are employing “reasonable methods” to ensure their enrollees have adequate access to providers. Consumer sources say the agencies’ push to regulate network adequacy is a step in the right direction, while an insurance source says industry worries that one of the pricing disclosure options will be difficult implement and is upset the administration didn’t extend reference pricing to the small group and individual markets as insurers had requested. Under the reference-based pricing strategy an insurer negotiates a price that it will pay for a certain service, and then requires an enrollee that chooses a higher-priced provider to pay the difference. The agencies’ Oct. 10 Frequently Asked Questions document states that reference pricing shouldn’t be used for emergency services. It also says plans must ensure an adequate number of providers accepting the reference price are available, create an easily accessible “exceptions” process for enrollees. disclose information on the reference prices free of charge, and upon request provide consumers a list of providers accepting the reference price for each service. Hospitals have long opposed reference pricing. A teaching hospital source says that academic hospitals will be unable to compete under the reference-priced structure due to their payer mix, and because of the high costs of training and offering other required services such as burn units and trauma centers. An earlier Frequently Asked Questions document issued by the three departments — HHS and Labor and Treasury departments — in May had said issuers need not apply cost-sharing to an enrollee’s out-of-pocket maximum if that individual uses a service that is more than the reference price, as long as the issuer or self-funded group plan provides “reasonable methods” to ensure adequate access to providers accepting set price. The maximum out-of-pocket (MOOP) spending for 2015 is $6,600 for an individual. The May FAQ sought comments on applying the MOOP to the reference-pricing design, and was particularly interested in what standards plans or issuers using reference-based pricing should be required to meet to ensure that individuals have meaningful access to medically appropriate, quality care. The agencies, in a new FAQ issued Friday, note that stakeholders sent in a range of comments and questions, with many suggesting that reference-price services be limited to non-emergency and routine care, and others recommending quality and network quality standards. Commenters also suggested that the plans be required to establish an exceptions 18 INSIDE CMS — www.InsideHealthPolicy.com — October 16, 2014 process and to disclose information on the pricing structure to consumers, the departments say. In general, Friday’s FAQ says that reference-based pricing that treats providers accepting the set price as the only innetwork providers should only apply to services in which the period between identification of the need for care and the provision of the care is long enough that consumers have time to make an informed choice. Reference pricing is therefore not reasonable for emergency services, the administration clarified. The FAQ also says plans must ensure that an adequate number of providers accepting the reference price are available. The departments provide no specifics on how to measure adequacy but say that plans should consider approaches developed by states, as well as geographic distance and patient wait times. Plans must also ensure that there are enough high-quality providers accepting the reference price. The FAQ further says that plans must create an easily accessible “exceptions” process for enrollees. This process must allow treatments from a provider not accepting the reference price to be considered as in-network in two cases: if there is no access to a provider accepting the reference price, or if the quality of the services could be comprised if the patient is required to go to the reference price provider. Finally, the FAQ says that plans must disclose information on the reference prices, free of charge, in two ways. First, plans must automatically provide information on the pricing structure, including a list of services and the price. Also, plans must provide to consumers, upon request, a list of providers accepting the price for each service; a list of providers accepting a negotiated price above the referenced price for each service; and information on the process and underlying data used to ensure that an adequate number of providers meet the quality standards. An insurance source says that the while plans are still reviewing the new FAQ, initial red flags stem from the requirement to provide information on reference-based pricing automatically since plans already have developed their materials for the 2015 plan year. Plans are also upset that the administration did not discuss extending reference pricing into the small group and individual market. Health expert Tim Jost of Washington and Lee University says that from a consumer perspective regulating group plan networks is a great idea, and the standards proposed are moving in the right direction. Jost, who is also a consumer representative to the National Association of Insurance Commissioners (NAIC), found the FAQ “fascinating,” saying that as far as he knows it is the first time the Department of Labor has attempted to impose network adequacy requirements on ERISA plans. Jost says he can see how DOL came to the decision, noting that if a plan says it will only pay a set price and everything above that price is not covered, it would “seriously undermine” the out-of-pocket limits if plans are not required to have reasonable networks. But, he says, it is a remarkable thing for the DOL to start telling ERISA plans what an appropriate network should look like. Kirsten Sloan, senior director of policy for the American Cancer Society Cancer Action Network, said in a emailed statement that the additional guidance on the use of reference-based pricing “takes important steps to protect consumers by requiring adequate disclosure to consumers, the availability of an exceptions process, and adequate number of providers accept the reference price.” “Given the complexities and uniqueness of cancer, the American Cancer Society Cancer Action Network (ACS CAN) recognizes that reference pricing may not be appropriate for cancer treatment. ACS CAN will continue to monitor the use of reference-based pricing and we are pleased the administration has committed to do so as well,” she adds. Hospital groups had slammed the administration for allowing plans to implement reference pricing, and said in their comments on the May FAQ that issuers should be required to apply cost-sharing to the ACA’s out-of-pocket limits. Atul Grover, of the Association of American Medical Colleges, said Friday that teaching hospitals oppose reference pricing because the facilities would be unable to compete at those prices due to the high number of Medicaid and uninsured patients treated at their hospitals as well as the extra facilities — such as burn units — that such hospitals are required to provided. If you turn healthcare into a commodity, all of the special things that the teaching hospitals provide would get zeroed out, says Grover. The FAQ does not address the question of who is responsible for supporting those critical services, he adds. —Amy Lotven Bayh: Chances of Device Tax Repeal ‘Very Good’ If GOP Wins Senate CHICAGO — There is a “better than 50/50” chance that Republicans will repeal the ACA’s device tax if they win the Senate in November, Evan Bayh, former Democratic Indiana senator and governor, said Tuesday (Oct. 7) at the Advanced Medical Technology Association’s medtech conference. A Republican-controlled Congress likely would first vote to repeal the Affordable Care Act, and after that measure is vetoed would settle on other changes to the law, such as a repeal of the industry-opposed excise tax, said Bayh, now a partner at McGuireWoods. The 2.3 percent excise tax was levied on medical device firms starting in January 2013. Device company CEOs said they have delayed hiring employees and scaled back research and capital projects, but those restrictions could be reversed with a repeal of the tax. Key lawmakers view repealing the tax as a priority and the president is not tied to the policy, Bayh said. If Democrats maintain a slim majority in the Senate a device tax repeal is still possible, he added. Under that scenario, INSIDE CMS — www.InsideHealthPolicy.com — October 16, 2014 19 many moderate senators could press for a device tax repeal in exchange for support of other measures in the final years of the Obama presidency. However, the chances of a repeal would be “very good” if Republicans win the majority, Bayh added. “If the Republicans don’t overreach on other things, I think this is doable,” he said. GOP members would likely try to fully repeal the ACA but the president would veto such a bill. Other changes, such as eliminating the employer mandate or repealing the device tax could be considered, and the president is not likely to issue a veto solely based on the medical device tax, he said. Finding an offset for the tax has been a challenge in advancing its repeal. But Bayh described the tax as a “rounding error” in terms of the federal budget. He noted there are “half of a dozen” items that could be looked at for savings. With political alignment, the issue of finding an offset could be resolved, he said. “If there’s the political will, they’ll find the fiscal way,” he said. “So generally you get asked this question now by people who really don’t want to do it; they’re just trying to put up an obstacle that could be overcome.” — Alaina Busch McBournie AARP Urges Congress To Extend CHIP Funding Through 2019 AARP called on Congress Tuesday (Oct. 14) to extend the Children’s Health Insurance Program funding — which is set to expire Sept. 30, 2015 — through 2019 as soon as possible in order to make sure that an estimated 2 million children don’t lose coverage and states’ budgets aren’t thrown out of whack due to a loss of federal dollars to the program. The nation’s largest seniors group, in a letter sent to leaders of the Senate Finance Committee and House Ways & Means and Energy & Commerce committees, notes this is an important issue for its members and older Americans, because the organization says grandparents across the country are raising an estimated 5.7 million grandchildren. While federal funding for the federal and state health insurance program for children of low income and working families is set to expire next year, most states are required to keep the program running through 2019 under a maintenance of effort requirement in the Affordable Care Act. CHIP was set to expire in 2019 because at that point families were expected to have health insurance either through Medicaid expansion, employer provided insurance or the health care exchanges. Since its inception in 1997 CHIP — which now provides health insurance to roughly 8 million children — has cut the percentage of children without insurance in this country in half, from 14 percent to 7 percent. “If Congress does not act, millions of children could either lose essential health coverage or need to enroll in individual, employer-based, or exchange plans with much higher out-of-pocket costs and less comprehensive benefits,” AARP writes. But should children lose coverage or see their coverage cut back because CHIP funding expires, AARP says, many would not be able to get coverage on the ACA health exchanges because of what is known as the “family glitch.” The glitch blocks spouses and children from access to exchange subsidies if a parent has an offer of coverage that is considered affordable for the individual, even if it is not affordable for the family. Sen. Jay Rockefeller (D-WV), the key architect of the CHIP program, has called on Congress to vote to extend funding in the lame-duck session following the November elections before he retires when his term is up in January. Rockefeller introduced legislation in June that would provide $88.7 billion to fund the program through 2019, but his office says that much of that spending would be offset. — Todd Allen Wilson CMS Pre-Pays Bonuses To Wire Up ACOs In Rural, Underserved Areas CMS has decided to loan money to ACOs in rural and underserved areas in response to complaints that some accountable care organizations can’t afford the information technology systems or the changes to their practices that are needed to perform well enough to receive bonuses. The initiative, unveiled Wednesday (Oct. 15), makes $114 million available to as many as 75 ACOs. CMS will recover those payments by paying less in shared-savings bonuses to ACOs that take out the loans, an agency release states. Both current ACOs and organizations that apply for the first year of the second round of ACO contracts in 2016 are eligible. The goal is to get providers to participate in areas where there has been little ACO activity — mostly rural providers — and to help existing ACOs take on the risk of pay cuts for missing cost and quality targets, CMS’ website states. There are three types of loans that organizations may apply for in 2016: up-front, fixed payment; payment based on the number of its prospectively beneficiaries; or monthly payments based on the number of prospectively beneficiaries. Existing ACOs may choose from two types of loans: up-front, variable payment based on the number of its prospectively-assigned beneficiaries; or monthly payments based on the size of the ACOs. Providers and policymakers have for a long time said ACOs work well in areas with big populations, but it is difficult for rural providers to set up the networks needed to be ACOs. Democrats have a lot riding on the success of ACOs, which is a key delivery reform under the ACA, and the administration is struggling to change the rules in a way that encourages participation while not cutting savings to Medicare by too much by paying out more bonuses and not penalizing poorperforming ACOs. — John Wilkerson 20 INSIDE CMS — www.InsideHealthPolicy.com — October 16, 2014
© Copyright 2026